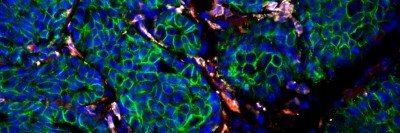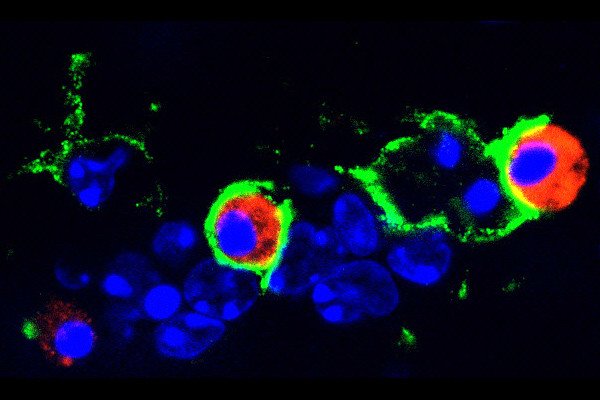
This image shows part of a glioblastoma tumor from a mouse treated with a CSF-1R inhibitor. The drug was shown to induce tumor-associated macrophages (green) to phagocytose or eat tumor cells (red).
White blood cells called macrophages patrol almost every tissue of the body to gobble up bacteria, dead cells, and other waste. About ten percent of the cells in the brain are macrophages, also known as microglia. These cells have many important functions such as protecting against infections and repairing damaged nerve tissue but have also been shown to promote brain diseases including Alzheimer’s disease, Parkinson’s disease, and multiple sclerosis.
Now a recent study led by Memorial Sloan Kettering scientists reveals that macrophages can support the growth and progression of glioblastoma brain tumors – the commonest and most deadly form of primary brain cancer – and that it might be possible to control the disease by manipulating these cells with a drug. The research was done in mice and published in the October issue of the journal Nature Medicine.
“Tumors are often infiltrated by macrophages and can ‘hijack’ these cells to spur their own growth,” explains Johanna Joyce, a member of the Sloan Kettering Institute’s Cancer Biology and Genetics Program and the senior author of the study. “Our findings suggest macrophages represent a potent therapeutic target in glioblastoma,” a disease that is exceptionally hard to treat with existing therapies.
Complex Tumors
Less than five percent of people with glioblastoma survive longer than five years after they are diagnosed even if they undergo intensive treatment with surgery, chemotherapy, or radiation. “There’s a crucial need for better strategies to control these aggressive tumors,” Dr. Joyce emphasizes.
So far scientists have had little success in developing targeted drugs against glioblastoma cells – drugs that home in on specific genetic mutations or biological pathways. “This is partly because glioblastoma tumors are very heterogeneous,” Dr. Joyce explains, referring to the fact that these tumors contain a highly diverse set of cancer cells with different genetic attributes.
In contrast, tumor-associated macrophages – which often make up as much as one third of the total tumor mass – tend to be more biologically uniform and could therefore be easier to target. Dr. Joyce and her colleagues looked at these cells in mouse models of a subtype of the disease called proneural glioblastoma. They noticed that the more advanced a tumor was, the more infiltrated by macrophages it tended to be.
“A similar pattern has been found when samples of tumor tissue from patients were analyzed,” Dr. Joyce says. “It seemed as if the tumor-infiltrating macrophages were helping the cancer advance.”
The investigators set out to test what would happen if the mouse proneural glioblastoma tumors were depleted of macrophages. They treated the mice with a drug that has been shown to eradicate macrophages in the brain and other organs. It works by inhibiting CSF-1R, a protein known to be essential for macrophage survival.
Surprise Findings
“There were a couple of surprises,” Dr. Joyce says. The first was how striking the drug’s effect was. It stopped newly formed tumors from progressing, caused more-established tumors to shrink, and made the mice live significantly longer.
The second surprise was no less positive, but initially confounding to the researchers. “We thought CSF-1R inhibition would wipe out the tumor-associated macrophages,” Dr. Joyce explains. “But when we looked at the tumors of mice who had been treated, the macrophages were still there” even though the drug had killed macrophages in the surrounding, normal brain tissue.
The researchers found that the macrophages residing in tumors had not succumbed to the drug because certain proteins secreted by the tumors were keeping them alive. But the therapy changed the macrophages’ behavior, blunting their tumor-promoting functions while making them more prone to elicit an anti-tumor response.
For example, these macrophages were induced to attack tumors by “eating” glioblastoma cells, a process known as phagocytosis.
“Our research suggests CSF-1R inhibition actually re-educates macrophages to attack tumor cells rather than support their growth,” Dr. Joyce says. “This is intriguing because if tumor-associated macrophages could be manipulated in the same way in the clinic – and be enlisted to actively fight a person’s cancer – this might ultimately be a more effective therapeutic strategy than to deplete the cells.”
Bringing Macrophage-Targeting Therapies to Patients
There could be several other advantages to developing glioblastoma drugs that act on macrophages and using these drugs in combination with existing therapies such as chemotherapy or radiation.
“Macrophages are genetically more stable than cancer cells, and therefore less likely to develop acquired drug resistance as glioblastoma cells often do,” Dr. Joyce explains. “In addition, we and other researchers have shown that targeting macrophages can increase the effectiveness of some chemotherapy drugs.”
CSF-1R inhibitors are currently being tested in early-stage clinical trials of glioblastoma patients and could be applicable in other diseases as well. “Studies have shown that in several cancer types, including breast, ovarian, thyroid, and pancreatic neuroendocrine tumors, increased numbers of tumor-associated macrophages correlate with poor patient prognosis, so it would be logical to test the drug in these diseases as well,” Dr. Joyce notes.