A common fungus called Aspergillus fumigatus can infect cancer patients with weakened immune systems and cause serious lung complications.
People with blood cancers such as leukemia or lymphoma are at a high risk of infections that can cause serious complications. This is especially true for patients receiving chemotherapy and bone marrow transplants. These treatments further weaken the immune system.
One of the biggest threats comes from a common fungus called Aspergillus fumigatus. It is carried through the air and is very difficult to avoid. Anyone who has seen mold grow on bread is familiar with it. A healthy person can inhale A. fumigatus with no ill effects because immune cells in the lungs are remarkably efficient at completely clearing infectious spores from the body.
But in someone whose immune system is compromised, even a single microscopic spore can readily spread to form filaments in the lungs, like branching spaghetti. This can wreak havoc on the body. An infection can destroy lung tissue and cause fever, cough, and discomfort when breathing. It can also spread to other sites in the body.
A research team led by Tobias Hohl, Chief of Memorial Sloan Kettering’s Infectious Diseases Service, and Amir Sharon of Tel Aviv University in Israel has found that immune cells in the lungs block these infections by inducing a form of programmed cell death, called apoptosis, in A. fumigatus. This finding, published in Science, could guide strategies for developing drugs that trigger this same type of cell death in the fungus, therefore preventing infections.
“We’ve identified a kind of Achilles’ heel of the fungus, and we would like to learn more about it so we can exploit that for therapeutic reasons,” Dr. Hohl says.
Neta Shlezinger, a postdoctoral fellow in Dr. Hohl’s lab, was the first author of the study. Collaborating on the research were Gerhard Braus of the University of Göttingen in Germany, and Robert Cramer of Dartmouth College.
Suicide Squads
Apoptosis has been well studied since the early 1970s. It plays an essential role in many biological processes and is used by many forms of life, from humans to fungi, to get rid of unneeded or abnormal cells. Under certain conditions, an enzyme called caspase induces specific cells to self-destruct for the benefit of the larger organism. This can involve cells dying off to sculpt the body during early development, but it is also a defense against pathogens.
For example, some immune cells that engulf a pathogen undergo apoptosis. This prevents the pathogen from reproducing and spreading beyond the initial site of infection. Some pathogens actually block this process. Then they can create a safe space for themselves so they can replicate before spreading.
Researchers knew that microbes could exploit the apoptotic pathways of more complex organisms in this way. They didn’t know, however, whether the reverse was true. Could higher organisms such as mice or humans exploit these same pathways in microbes?
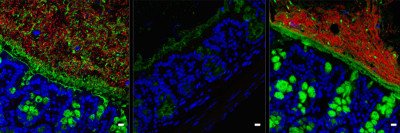
Dr. Hohl’s team suspected that this might be the key to how the lung’s immune cells ward off infection from A. fumigatus. To investigate this, his lab developed a method of coupling A. fumigatus to fluorescent molecules that signal whether the fungus is alive or dead.
“This technology enables us to probe the outcomes of interactions between individual fungal cells and immune cells in a live organism, such as a mouse,” he says. “It allows us to visualize the apoptotic process in the fungal cells and understand how it unfolds.”
By analyzing lung tissue from mice infected with the fluorescent A. fumigatus, the researchers determined that the microbes were taken in by immune cells in the lungs called neutrophils and destroyed. They observed this process in human neutrophils as well. But were the neutrophils actually triggering apoptosis in the fungus or using some other method?
“We knew that the fungus was dying, so it wasn’t an enormous stretch to think that apoptosis might be occurring,” Dr. Hohl says. “But it wasn’t clear whether this was the case or how important apoptosis was to overall protection from infection.”
Jamming the Signal
The researchers were able to identify a gene in the fungal cells called AfBIR1. It acts as a brake to suppress the caspase enzyme from activating and triggering apoptosis. When mice were given A. fumigatus that had been genetically modified to express AfBIR1 at high levels, the fungus survived and the mice succumbed to the infection. When the researchers dampened the effect of AfBIR1 using a drug, the fungus underwent apoptosis more readily and was quickly wiped out. This provides strong evidence that apoptosis and the level of AfBIR1 expression are critical factors in a healthy person’s defense against A. fumigatus infection.
Another important element — the strength of the immune defense — also influences the risk of infection. The researchers found that neutrophils normally create a condition called oxidative stress to kill A. fumigatus. Mice (and presumably people) with weakened immune systems cannot subject the fungus to oxidative stress. Under these conditions, AfBIR1 expression is sufficient to block apoptosis. This enables the fungus to persist.
Dr. Hohl says that the next step is to tease apart the components of the apoptosis pathway in A. fumigatus and then develop and test different drugs that might induce apoptosis, possibly by targeting AfBIR1. Existing antifungal drugs target the fungus’s ability to build a cell wall. Complementing them with a drug that provokes apoptosis could make these therapies much more effective.
“If we could figure out ways to trigger apoptosis in fungal cells artificially, with a lesser need for immune cells, that would be a very novel way to address this major complication of cancer treatment,” he says.