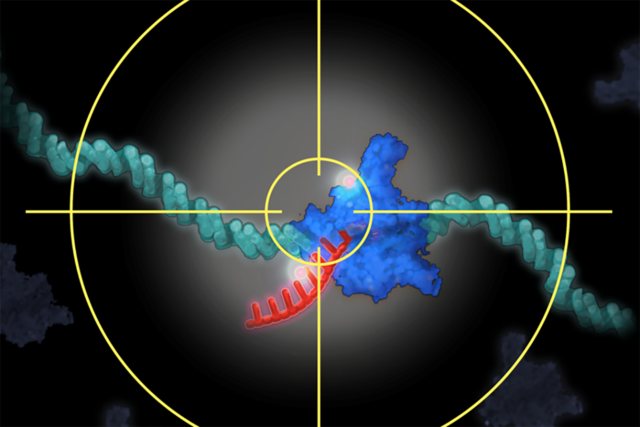
Visualizing individual molecules at specific sites inside cells is a challenging task for structural biologists. Now MSK researchers have developed a system that can zoom into a precise location in a cell’s nucleus. From there, they can selectively illuminate a single gene and its activity. This illustration of the concept depicts the system locked onto a single gene and the molecules that interact with it. Illustration by Terry Helms.
A picture is worth a thousand words, or so the saying goes. But it can be quite a challenge to capture a picture of something that’s so tiny it’s on the scale of individual molecules.
The field of structural biology is dedicated to constructing images of very, very small things. But most of the techniques used by structural biologists to take these pictures require that the molecules are frozen in one position. This makes it difficult to watch the dynamic, shifting processes that are essential to life.
For the first time, researchers from the Sloan Kettering Institute have found a way to peer inside living cells and observe gene transcription. This is the process by which DNA is copied into messenger RNA (mRNA), which then specifies how a protein is made.
“Gene transcription is one of the most fundamental processes in all of biology,” says SKI structural biologist Alexandros Pertsinidis, senior author of the study, which was published in Cell. “We know that it’s highly regulated and uses complicated molecular machinery. Being able to watch this process as it happens is an important step forward in understanding what goes on inside cells.”
Following the Recipe
If you think of the genetic code as a universal cookbook, containing all of the instructions needed to make every part of a living organism, you can think of the various cell types as different restaurants, Dr. Pertsinidis explains. “French restaurants follow French recipes to make French dishes, and Italian restaurants follow Italian recipes to make Italian dishes,” he says. “In the same way, brain cells make the proteins that brain cells need to function, and liver cells make the proteins for liver function.”
A family of enzymes called RNA polymerases and a large set of factors that are associated with them regulate the transcription of individual genes and control the characteristics that cells exhibit. “The interplay between RNA polymerases and regulatory factors helps determine which genes are turned on and off in specific cells,” he says. “They also control how cells respond to outside signals, which can influence their activities.”
Until now, the function of RNA polymerases and associated regulatory factors has been studied indirectly, through biochemical reactions: Cells are broken open in a test tube and purified into individual parts. By adding or removing components and measuring the outcomes, scientists have been able to figure out certain molecular activities. How the machine as a whole works inside cells, however, has remained obscure.
“For 50 years, hundreds of researchers all over the world have studied these reactions,” Dr. Pertsinidis says. “But the problem has been that nobody has been able to directly observe how gene transcription happens inside a live cell.”
Zooming In on a Single Gene
In the study, the investigators used a highly specialized optical microscope to look at the activity of one RNA polymerase, called RNA polymerase II, as it interacted with genes and synthesized mRNA. The new method, developed by Dr. Pertsinidis’s lab, is called single-molecule nanoscopy.

To be able to look at the individual parts of cells, researchers label molecules with a fluorescent tag that makes them glow under the microscope. “But a cell is very crowded, and there are many reactions happening at the same time,” Dr. Pertsinidis says. “If you label all the polymerases in a cell, the whole nucleus is just a big glow.
“What’s new about this technology is the ultrasensitive, integrated system that lets us zoom in on a single tagged gene even when the cell nucleus is moving around and the specific chromosomal location is jiggling due to random microscopic motion,” he says. “At the same time, the system suppresses the signals from the other reactions that are happening, casting them into the background. This enables us to extract the signal for only the gene of interest and zoom in on it.”
The organization and dynamics of RNA polymerase II in the nucleus have been a topic of intense study over the past few decades. “Here, we directly observed the activity of this molecule and how it functions in the nucleus of live cells,” Dr. Pertsinidis says. “Being able to see how it interacts with other regulatory factors has unveiled the intricate hierarchies and interdependencies of these various factors. These insights enable us to reach a more detailed and comprehensive picture of transcription in live cells.”
Expanding to Other Cellular Processes
The researchers hope that their tool will be widely used to study complicated reactions inside living cells.
“There are enough details in our paper that other labs will be able to pick up and implement the technology,” Dr. Pertsinidis says. “We also have labs both inside and outside MSK that are interested in collaborating with us on specific projects.”
He adds that although he is focused on understanding gene transcription, the tools his team has developed could be used to study the details of other vital biological processes, such as DNA repair and protein synthesis.




