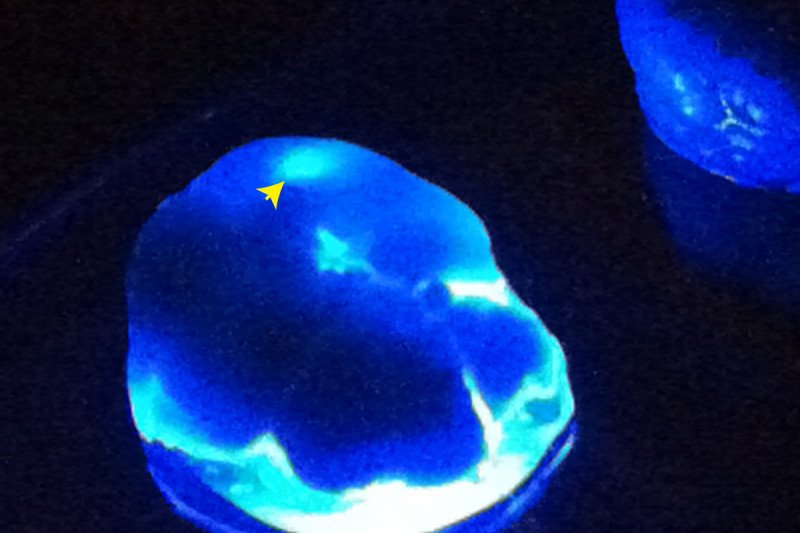
Fluorescence image of an experimental brain tumor (indicated by arrow) originating from iPS cells.
Regenerative medicine is based on the idea that stem cells could be used to regrow other cells or tissues that have been lost due to injury or disease. Although the field has shown much promise in animal studies and even a few early-stage clinical trials, a potential problem in the technology is that these engineered stem cells can grow too well and lead to cancer.
A multidisciplinary team from Memorial Sloan Kettering has tackled this problem and come up with a possible solution. Led by research fellow Elisa Oricchio, the scientists introduced a gene into the reprogrammed cells that could be switched off if needed, killing the engineered cells that had become cancerous while allowing the healthy ones to continue growing.
“There are many centers that are developing engineered stem cells for use in the clinic, for treatment of a variety of diseases,” says MSK cancer biologist Hans-Guido Wendel, senior author of a study being published today in Cell Reports. “When you are doing this type of therapy, you really need a fail-safe method to prevent the development of tumors, and that’s where this technology will be useful.”
Induced Pluripotent Stem Cells
The research builds on techniques developed to convert adult cells such as skin cells into induced pluripotent stem (iPS) cells. These iPS cells have the potential to form almost any cell type in the human body — including nerve, pancreas, or liver cells, for example — and are genetically matched to the patient they are being used to treat.
With just a few exceptions, such treatments are still the stuff of science fiction. However, these techniques have been studied widely in animal models, especially mice. And researchers have discovered something alarming: Up to 20 percent of these engineered cells turn cancerous in animal models.
In the current study, the particular therapeutic cells used were iPS cells that had been programmed to form dopamine neurons, the type of brain cells that are lost in Parkinson’s disease (PD). MSK developmental biologist Lorenz Studer, who was a coauthor on the Cell Reports paper, has been developing and refining these therapeutic cells for the treatment of PD for more than a decade.
“The risk of tumor formation remains an important concern for bringing iPS cell–based therapies to the patient,” he says. “We have worked very hard on eliminating any tumor-forming cells in our Parkinson’s project. This new study offers an elegant fail-safe mechanism to further increase safety.”
Finding the Right Switch
The gene used as a kill switch in the current research, called OMOMYC, is an inhibitor of a major cancer gene called MYC. The investigators engineered the OMOMYC gene into the dopamine neurons, along with a reporter gene that causes tumor cells to glow, making them easy to detect in laboratory mice.
After inserting the engineered dopamine neuron cells into the brains of the mice, the investigators found that some of the animals developed brain tumors. They then showed that when the MYC gene was turned off, which was done by giving the mice a drug, tumors were induced to shrink. At the same time, normal neurons derived from the stem cells were not affected. This indicated that OMOMYC can selectively kill the cancerous cells without touching the newly made neurons.
“It turns out that normal differentiated cells don’t care if you switch MYC off. In fact, they express only very low levels of it, and it appears they don’t really need it,” Dr. Wendel says. “But when you switch it off in these malignant brain tumors, they stop growing and just freeze.”
“The idea of inserting a suicide gene has been around for a while in regenerative medicine,” he adds. “The catch is that if you’ve generated new cells that are treating someone’s disease, you don’t want to destroy them. The difference is that we can spare the healthy cells and at the same time effectively kill cancer cells. Potentially, this gene can be engineered into stem cells and provide us with a powerful fail-safe that we can activate when we need it.”
Applying the Research to Other Cell Types
The MSK team is currently adding the OMOMYC kill switch to other types of engineered cells to test the effects. For example, one application may be T cells that are designed to treat certain types of leukemia and other cancers. These T cells are already in a handful of clinical trials at MSK.
“The use of cell therapies to treat cancer through immunotherapy, genetic disorders through gene therapy, and degenerative or age-related disorders through regenerative medicine will be sharply increasing over the next decade,” says Michel Sadelain, Director of MSK’s Center for Cell Engineering and another coauthor on the study. “The availability of a safety switch that spares therapeutic cells and eliminates those that are cancerous is a great asset for the field.”





