
Computational biologist Joao Xavier (left) and Eric Pamer, Chief of the Infectious Diseases Service
At first glance, tiny single-celled bacteria could not be more different from humans. But there are many reasons why studying them can teach us a lot about human health, and about cancer in particular.
Bacterial infections can cause major complications for cancer patients. At Memorial Sloan Kettering, scientists studying bacteria not only learn about infections but also gain new insights about cancer. Studying how bacteria evolve to develop resistance to antibiotics — a phenomenon that has been recognized for several decades — can be a model for studying how tumor cells develop resistance to newer targeted therapy drugs for cancer.
And as we learn more about how the immune system can be harnessed to fight cancer, it is instructive to understand in greater detail how its components recognize and attack infections by bacteria.
On the laboratory side, bacteria have been model organisms for studying the control and regulation of genes since the birth of modern genetics in the 1950s. Recent advances in our understanding of cancer genetics would not be possible without the foundation built by studies of bacterial genes.
More recently, the Human Micro-biome Project, a multi-institutional initiative funded by the National Institutes of Health, has aimed to characterize all of the microorganisms colonizing the human body. The project, which has cataloged the normal microbes (known as the microbiota) inhabiting healthy humans, has offered the most detailed insight to date into the normal variations and complexities of these commensal microorganisms.
One of the ways that Memorial Sloan Kettering has committed to the study of bacteria is through the creation of its Lucille Castori Center for Microbes, Inflammation, and Cancer. The Castori Center was established in 2010 to shed light on the role that microbes and the body’s inflammatory and immunological responses to them play in the development of cancer.
“The Castori Center brings together the many investigators at Memorial Sloan Kettering who are focusing on various aspects of microbial infections,” explains Eric G. Pamer, who leads the Castori Center and is also Chief of the Infectious Diseases Service and a researcher in the Sloan Kettering Institute’s Immunology Program. “Memorial Sloan Kettering is uniquely positioned to investigate these types of issues.”
Some of Dr. Pamer’s own work has looked at the role that antibiotics play in increasing susceptibility to infection with certain types of bacteria, a common complication in hospitalized patients. He has studied how antibiotics given to patients during the course of treatment to either relieve or prevent infections may have a negative effect on the good or health-promoting bacteria that live in the body, allowing harmful bacteria to dominate.
What follows are three examples of how the study of bacteria informs cancer research at Memorial Sloan Kettering.
Drawing Parallels between Tumor Evolution and the Social Lives of Bacteria
Computational biologist Joao Xavier has spent most of his career researching how bacterial cells interact with one another and produce complex group behaviors. He had never studied cancer before joining Memorial Sloan Kettering in 2009. “I came here partly because I was intrigued by cancer biology,” he says. “I kept seeing all these parallels between microbial communities and tumors that I wanted to explore.”
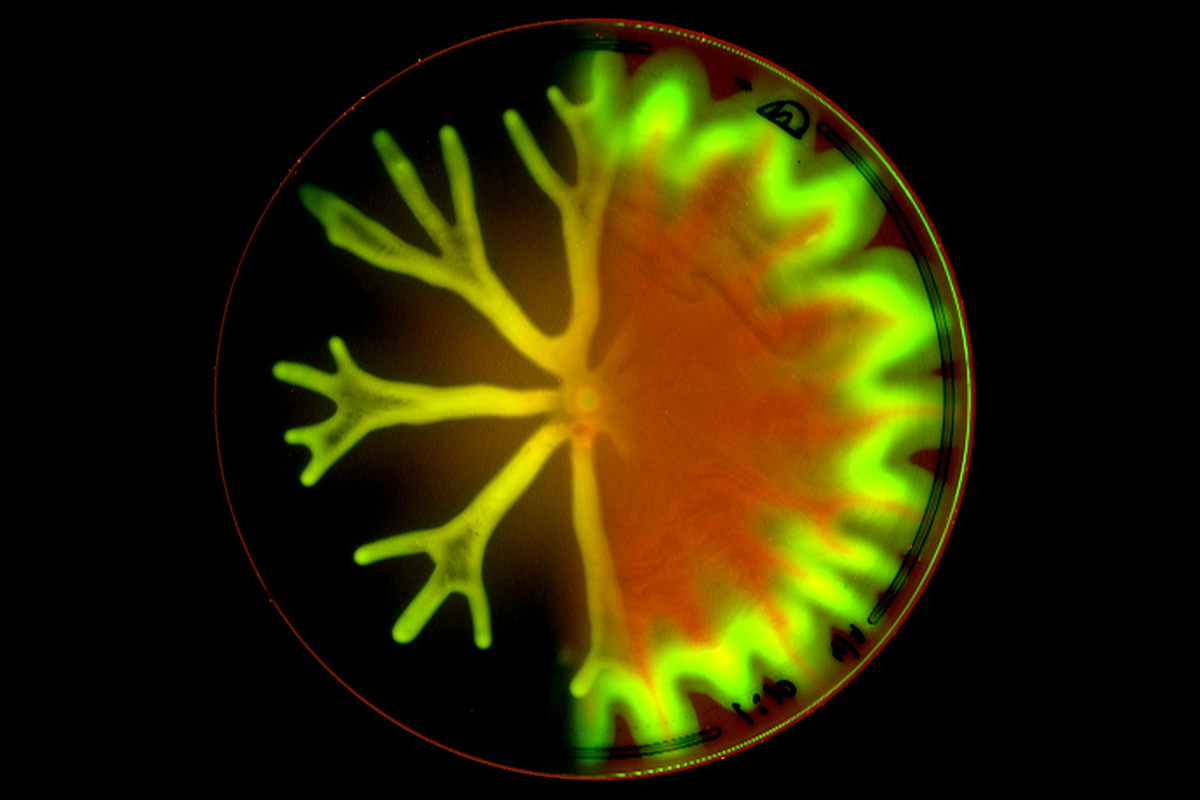
This image from the lab of Joao Xavier shows Pseudomonas aeruginosa, a common bacterium, producing a complex group behavior known as hyperswarming, which causes colonies of cells on the right to migrate quicker across a petri dish. The lab now applies computational methods originally developed for bacterial communities to study the biology of tumors.
Cancer cells and bacteria are different in many ways, but common principles hold for how populations of cells behave and evolve. As mentioned previously, the emergence of drug resistance is one example. Dr. Xavier explains that in the same way antibiotic treatment of an infection can create strong evolutionary pressure that favors the survival of drug-resistant bacteria, so can treatment with some cancer drugs select for clones of cells within a tumor that are able to grow in spite of the drug, which eventually renders the tumor unresponsive to it. “Whether we are interested in drug resistance or other aspects of cancer biology, we can learn a lot about tumors at the conceptual level by studying bacterial evolution,” he says.
Bacteria commonly grow in densely packed communities known as biofilms, aggregates of cells embedded in a gluey matrix. Dr. Xavier’s team has developed computer models to analyze how cells within biofilms interact chemically and biophysically, and has combined this computational research with laboratory experiments. Recently they applied a similar system to study how cancer cells within a tumor interact with noncancerous cells known as macrophages.
A macrophage is a type of cell that inhabits many different organs and forms an essential part of the body’s line of defense against pathogens. In people with cancer, however, macrophages often start growing inside and around a tumor, and their normal function can be subverted to help the tumor grow and metastasize.
The work was a multidisciplinary collaboration led by Dr. Xavier, post-doctoral fellow Carlos Carmona Fontaine, and Sloan Kettering Institute cancer biologist Johanna Joyce. Together, the researchers set out to explore the spatial patterns of macrophages in tumors. “When a tumor is small, it’s uniformly infiltrated by macrophages,” Dr. Xavier explains. “But as it grows, macrophages tend to stay on the outside” of the tumor, possibly shielding the cancer cells within from detrimental cells and factors in the nearby environment. “This pattern had been observed in many tumor types, but to our knowledge no one had looked at why it forms,” he says.
He and his colleagues used the same computer models they use for research on bacterial biofilms to make predictions about the complex interplay of cancer cells and macrophages and tested these predictions in experiments with cells growing in culture. Ultimately their research showed that the redistribution of macrophages to the tumor periphery is driven by metabolic changes in tumor cells that lead to high levels of lactic acid, making the tumor’s interior toxic to macrophages. Over time, macrophages stay at the tumor’s periphery, where they have better access to nutrients.
Dr. Xavier thinks the research, which was published in the journal PNAS last year, may inspire more scientists to consider how changes in cancer cell metabolism affect noncancerous cells in the tumor microenvironment. In addition, the findings could open possibilities for the development of new drugs that act on tumor-associated macrophages rather than on cancer cells.
“Our research is only a small piece in this puzzle,” Dr. Xavier says. “But it’s a unique piece that we were able to contribute perhaps in part because we’re not cancer biologists and came to the problem with an outside perspective.”
Immune Stimulation and Unexpected Paths
A noteworthy feature of bacteria is their capacity to stimulate the immune system to attack tumors. This concept was first put to therapeutic use in the 1890s, when William Coley, a surgeon at New York Cancer Hospital (the predecessor to Memorial Sloan Kettering Cancer Center), developed Coley’s toxins, a cocktail of killed bacteria injected into tumors that sometimes brought complete remission.

Michael Glickman (left) and Gil Redelman-Sidi.
Coley’s toxins were used against various cancers, and, because they were highly effective in rare circumstances, it stimulated laboratory investigations that ultimately led to much of modern cancer immunotherapy.
One bacterial therapy, however, is still a standard treatment for early-stage bladder cancer. In the 1950s, Memorial Sloan Kettering cancer immunologist Lloyd J. Old and other researchers began investigating the antitumor effects of BCG (bacillus Calmette-Guérin), a weakened form of a bacterial pathogen that had been used for decades as a vaccine for tuberculosis. Clinical studies conducted at Memorial Sloan Kettering demonstrated that BCG therapy prevents bladder cancer from returning after removal of a tumor.
Researchers suspect BCG incites some form of immune response against tumors, but the exact mechanism is unclear. In 2013, Memorial Sloan Kettering physician-scientist Michael S. Glickman and infectious disease specialist Gil Redelman-Sidi reported in the journal Cancer Research that the bladder cancer cells themselves may play a role. They discovered that some of the same genetic mutations that cause bladder cells to become cancerous also activate a process that allows BCG to enter the cell.
Two of the known mutations, in the genes RAS and PTEN, are implicated in a number of other cancers. (The identification of PTEN’s role came through a collaboration between the Glickman lab and the laboratory of Memorial Sloan Kettering cell biologist Xuejun Jiang.)
Dr. Glickman’s laboratory is now collaborating with urologic surgeon Bernard H. Bochner to explore whether analyzing a patient’s bladder cancer cells can reliably predict responsiveness to BCG therapy. With support from The Society of Memorial Sloan Kettering and the Lucille Castori Center, Dr. Redelman-Sidi is developing a lab test that would analyze urine samples — which usually contain cancer cells shed from the bladder — to predict whether BCG therapy would be the best course of treatment.
The researchers are now testing genetically modified BCG strains that would be even more effective against bladder cancer, as well as less prone to infecting normal cells — a side effect that sometimes occurs.
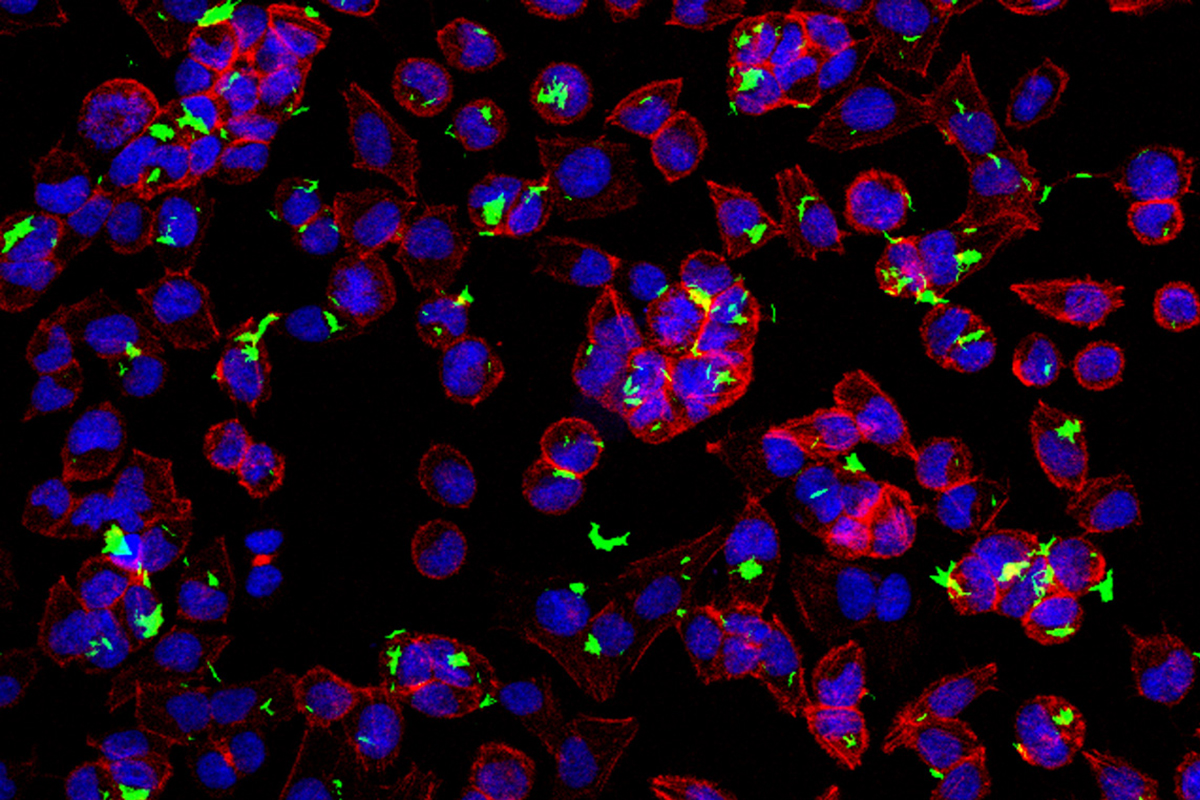
The image shows BCG (green) infecting bladder cancer cells (blue and red).
In addition to discovering the role of RAS and PTEN mutations in allowing BCG entry, the Cancer Research paper also described the unusual pathway taken by the pathogen.
“BCG is a mycobacterium, usually taken up only by certain immune cells looking for invaders to destroy,” Dr. Glickman explains. “BCG is not equipped with a means to force itself into other types of cells such as cancer cells. But we learned that BCG enters cancer cells by another pathway called macropinocytosis, which is activated by the RAS and PTEN mutations.”
The insights about BCG’s reliance on macropinocytosis could have broader implications for cancer therapy.
“When Gil and I made the finding about macropinocytosis, there were parallel discoveries by others showing that the same pathway of macropinocytosis is critical for RAS-mutated cancer cells to take up nutrients,” Dr. Glickman says. “We now have a collaboration with [Memorial Sloan Kettering President and CEO] Craig Thompson investigating this pathway and its role in fueling the growth of RAS-transformed cancers — which are numerous and very difficult to treat.”
Dr. Glickman says this type of crossover illustrates the advantage of studying bacteria in a cancer-centered environment. “Much of what we do in my lab is basic tuberculosis biology, but in this case our investigations in trying to understand bacterial therapy for cancer provided insight into a more fundamental cancer biology process.”
Preventing Dangerous Side Effects of Transplants
Memorial Sloan Kettering physician-scientist Robert Jenq is studying the role of bacteria in graft-versus-host disease (GVHD), a major complication of allogeneic bone marrow or stem cell transplants, in which patients receive cells from donors. GVHD occurs when newly transplanted immune cells from the donor recognize the recipient’s tissue as foreign and attack it. The potentially fatal condition can result in damage to the liver, lungs, digestive tract, or other organs.

Hematologic Oncology Division Head Marcel van den Brink (left) and physician-scientist Robert Jenq
Dr. Jenq’s particular research is focused on the role of bacteria and how certain bacterial strains that are naturally found in the body may protect against GVHD.
“Since the 1970s, we’ve known that the microbiota have an impact on the risk for GVHD,” he says. “We tried to apply some of the early discoveries we made in mouse models to patients, but in the end they didn’t work.”
“Now new DNA sequencing technologies have helped us to characterize all the bacteria that live in the gastrointestinal tract in great detail,” Dr. Jenq explains. “This approach is something that wasn’t possible even a few years ago.”
Previous studies from Dr. Jenq and his colleagues, including Hematologic Oncology Division Head Marcel van den Brink, began looking at how the balance of intestinal bacteria can affect GVHD.
A recent study from Dr. Jenq, done in collaboration with Dr. van den Brink and Dr. Pamer, as well as infectious disease specialist Ying Taur, focused on a genus of bacteria called Blautia. “Using sequencing technologies to study the bacterial makeup of the gastrointestinal tracts of patients undergoing bone marrow transplants [BMTs], we found that if patients had even a smidgen of Blautia left — just 0.1 percent of the total makeup of bacteria in their GI tract was Blautia — they had an almost negligible risk of GVHD,” Dr. Jenq says.
The investigators took their findings back to mouse models and found that giving Blautia to mice that had lost it could protect them against GVHD.
“Blautia is a harmless type of bacteria that every patient comes in with, but a lot of them lose it during the course of transplantation,” he adds. “It seems to be sensitive to the antibiotics that patients are given during the course of treatment, and it also declines in patients who aren’t eating, a common situation in BMT patients.”
Currently, GVHD is treated with powerful immune suppressants. “It’s necsary because GVHD can be fatal,” Dr. Jenq says, “but these drugs increase the risk that patients will develop infections and also that their cancer may come back. Our findings about Blautia open up new avenues for preventing GVHD.”
Memorial Sloan Kettering clinicians are considering a number of different approaches, which include treating patients with antibiotics that do not harm their Blautia colonies and finding ways to maintain their beneficial bacteria after they have received a transplant. Encouraging patients to eat during their transplant — rather than providing intravenous nutrition — may have major benefits. “When you supply nutrients by IV, you’re feeding the patients, but you’re not feeding their bacteria,” Dr. Jenq notes. “That may be important.”
Another approach could be finding a way to give patients beneficial bacteria such as Blautia after their transplants. (This is the basis of the idea behind the food supplements known as probiotics.)
Some studies have suggested that the role of Blautia in the gastrointestinal tract is to produce compounds called short-chain fatty acids, which researchers believe help to suppress inflammation. “If this proves to be true, we may be able to bypass the bacteria completely and just give as a treatment the compounds the bacteria are producing that mediate the beneficial effects,” he says. “We call this strategy ‘postbiotics.’”
All of these possibilities need to be evaluated further before any clinical studies can begin in patients. “Testing all of these strategies in mice will shape whatever trials we do in the future,” Dr. Jenq concludes.


