Cancer Biology & Genetics Program
The Richard White Lab
Research

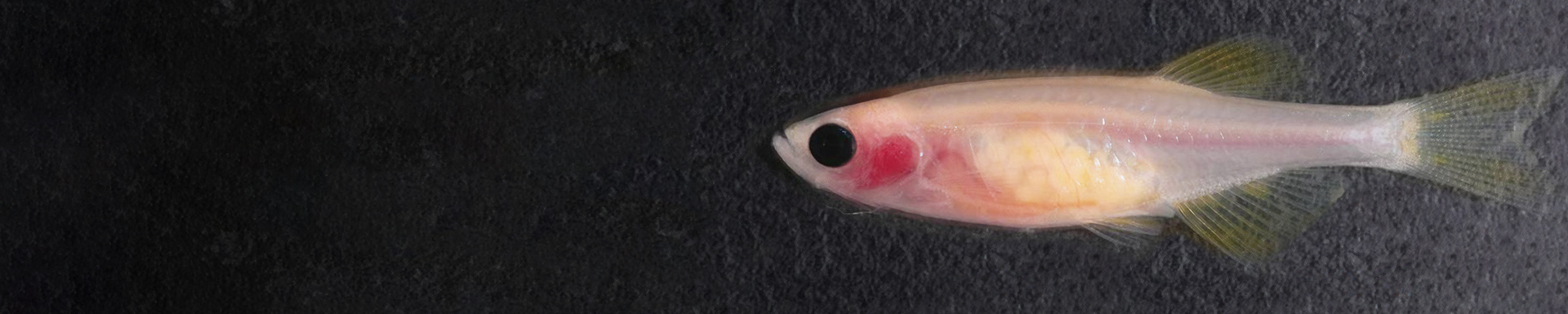
Zebrafish models of cancer
We are interested in the ways in which developmental programs affect cancer progression, and how these programs are altered by the microenvironment. To address these questions, we primarily utilize the zebrafish as a model, given its strengths in genetic manipulation and imaging, which is enhanced in our previously developed transparent casper strain of fish. We additionally use human pluripotent stem cell models of cancer to complement what can be done in vivo in the fish. With state-of-the-art genetic engineering and high-resolution imaging, our goal is to dissect these interactions to understand how tumors start and eventually metastasize to new locations.

Featured News


Publications Highlights
Baggiolini A+, Callahan SJ+, Montal E, Weiss JM, Trieu T, Tagore MM, Tischfield SE, Walsh RM, Suresh S, Fan Y, Campbell NR, Perlee SC, Saurat N, Hunter MV, Simon-Vermot T, Huang TH, Ma Y, Hollmann T, Tickoo SK, Taylor BS, Khurana E, Koche RP, Studer L*, White RM* (2021). +co-authors, *co-corresponding authors. Developmental chromatin programs determine oncogenic competence in melanoma. Science 373 (1104): abc1048. PMCID pending.
Weiss JM, Hunter MV, Tagore M, Ma Y, Misale S, Simon-Vermot T, Campbell NR, Newell F, Wilmott JS, Johansson PA, Thompson JF, Long GV, Pearson JV, Mann GJ, Scolyer RA, Waddell N, Montal ED, Huang T, Jonsson P, Donoghue MTA, Harris CC, Taylor BS, Ariyan CE, Solit DB, Wolchok JD, Merghoub T, Rosen N, Lezcano-Lopez C, Hayward NK, White RM (2021). Anatomic position determines oncogenic specificity in melanoma. in revision, available at: https://www.biorxiv.org/content/10.1101/2020.11.14.383083v1
Campbell NR, Rao A, Hunter MV, Sznurkowska MK, Briker L, Zhang M, Baron M, Heilman S, Deforet M, Kenny C, Ferretti LP, Huang TH, Perlee SC, Garg M, Nsengimana J, Saini M, Montal E, Tagore M, Newton-Bishop J, Middleton MR, Corrie P, Adams DJ, Rabbie R, Aceto N, Levesque MP, Cornell RA, Yanai I, Xavier JB*, White RM* (2021). *co-corresponding authors. Cell state diversity promotes metastasis through heterotypic cluster formation in melanoma. Developmental Cell, 2021 Sep 13:S1534-5807(21)00677-8
Hunter MV+, Moncada R+, Weiss JM, Yanai I*, White RM* (2021). +co-authors, *co-corresponding authors. Spatial transcriptomics reveals the architecture of the tumor/microenvironment interface, Nature Communications, in press, available at https://www.biorxiv.org/content/10.1101/2020.11.05.368753v1
People

Richard White, MD, PhD
- Cancer biologist and oncologist Richard White uses the zebrafish to dissect interactions between tumor cells and the microenvironment that promote metastasis.
- Albany Medical College: M.D., Ph.D.
- Yale New Haven Hospital: Internal Medicine Residency & Chief Residency
- Dana Farber Cancer Institute & Massachusetts General Hospital: Medical Oncology Fellowship
- Children’s Hospital Boston & Harvard Medical School: Postdoctoral Fellowship
- [email protected]
- Email Address
Members














Senior Research Scientist

Research Fellow

Postdoctoral Fellow

Postdoctoral Fellow

Graduate Student

Assistant

MD-PhD student

Graduate Student

Research Fellow

Graduate Student

Research Fellow

Research Fellow

Research Technician

- Princeton University



- BS, Massachusetts Institute of Technology


- University of Texas Austin



Lab Alumni

Vice President, Director of Medical Strategy at Havas Health & You, New York, NY

Clinical Fellow

MD–PhD student

Assistant

Senior Research Technician

Research Technician

Graduate Student

Postdoctoral Fellow

Postdoctoral Fellow

Graduate Student

Senior Technician

Graduate Student


MD, PhD Student


Graduate Student

Lab Affiliations
Achievements
- Josie Robertson Investigator (2012-2017)
- NIH Directors New Innovator Award
- Young Mentor Award, Melanoma Research Alliance
- Young Investigator Award, American Association for Cancer Research/American Society of Clinical Oncology
- Young Mentor Award, Harvard Medical School
Read more
- Outstanding Educator Award, Yale School of Medicine, Department of Internal Medicine
Open Positions
To learn more about available postdoctoral opportunities, please visit our Career Center
To learn more about compensation and benefits for postdoctoral researchers at MSK, please visit Resources for Postdocs
Get in Touch
-
Lab Head Email


