Common Names
- Squalamine lactate
For Patients & Caregivers
Tell your healthcare providers about any dietary supplements you’re taking, such as herbs, vitamins, minerals, and natural or home remedies. This will help them manage your care and keep you safe.
Squalamine may have anticancer effects, but definitive evidence is lacking.
Squalamine is a compound derived from dogfish shark tissues. Lab studies suggest it can kill bacteria and block growth of new blood vessels.
Small clinical studies suggest that squalamine is safe and well tolerated when given to patients with cancer and age-related macular degeneration. However, these studies used an injectable form, and it is unclear if oral squalamine products have the same effects. Further research is needed.
Squalamine should not be confused with squalene, an oil found in shark liver.
- To treat cancer
Small clinical studies have shown that the injectable form of squalamine is safe in patients with solid tumors, but it is not known if it can treat cancer. - To treat age-related macular degeneration
Small clinical studies suggest that squalamine is safe and well tolerated, but it is not known if it can be used to treat this condition. - To treat bacterial infections
Squalamine showed antibacterial effects in lab studies, but clinical trials have not been conducted.
- Fatigue, nausea, anorexia, and liver toxicity with intravenous squalamine in a clinical study.
For Healthcare Professionals
Squalamine is a water-soluble steroidal antibiotic first identified in dogfish shark (Squalus acanthias) tissue (1). It is now produced synthetically, and has significant bactericidal and fungicidal effects. It also enhances bactericidal effects when used in combination with standard antibiotics (2) (3) (4), and significantly reduces aggregation of alpha-synuclein, a protein associated with Parkinson’s disease and related syndromes (18).
Preclinical studies show that squalamine inhibits growth factor-dependent pathways in endothelial cells. It has been studied since as a treatment for age-related macular degeneration and cancer (19) (20), with the injectable form appearing to be well tolerated in early-phase trials (5) (6) (7). However, it is not known if oral squalamine would have similar effects.
Other preliminary findings suggest that topical squalamine along with ranibizumab improves visual recovery in patients with retinal vein occlusion-related macular edema (21), and a squalamine ointment demonstrated partial clinical activity against ringworm (22).
Squalamine analogues are currently being studied for nasal decolonization of Staphyloccus aureus to prevent risk of associated infections (23), and squalamine eye drops are being tested in the search for novel treatments of neovascular age-related macular degeneration (24).
Squalamine should not be confused with squalene, an oil found in shark liver.
- Dogfish shark, Squalus acanthias: Squalamine is found primarily in the liver and gallbladder, but also the spleen, testes, stomach, gills, and intestine.
- Sea lamprey, Petromyzon marinus (2)
- Cancer treatment
- Age-related macular degeneration
- Bacterial infections
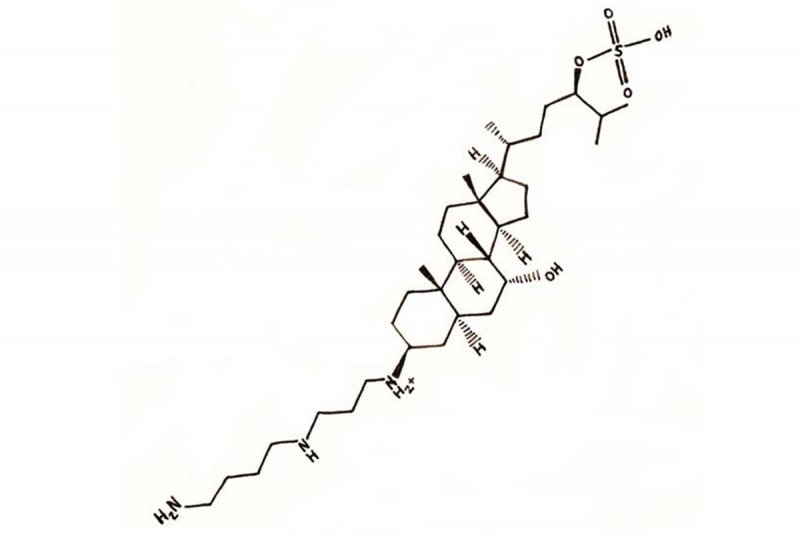
Squalamine is a cholestane steroid conjugated to a spermidine at position C-3. However, it does not have glucocorticoid and mineralocorticoid effects (8). Squalamine binds to cell membranes and inhibits the specific membrane Na+/H+ exchanger NHE3, causing intracellular pH alterations and disruption of intracellular signaling induced by angiogenic growth factors (8).
Squalamine alters shape and volume of endothelial cells in embryonic vascular beds, causing narrowing of the vessel lumen and occluding blood flow (10). These actions inhibit multiple key steps in angiogenesis, including mitogen-induced actin polymerization, cell-cell adhesion, and cell migration, ultimately inhibiting endothelial cell proliferation. Squalamine blocks downstream VEGF signaling pathways, including phosphorylation of p44/p42 MAP kinase in vascular endothelial cells (11). It also disrupts F-actin fibers and induces internalization of vascular endothelial-cadherin from the membrane into the intracellular compartment (12). It may also decrease retinal neovascularization that is thought to encourage macular degeneration (17).
Squalamine is an amphipathic compound that interacts with various membrane glycerophospholipids (4). It has a faster killing rate of gram-positive versus -negative bacteria (3) (13) (14). It also enhances chemotherapy drug cytotoxicity by promoting tumor cell apoptosis and reducing angiogenesis (10) (15) (16). Inhibition of growth factor-induced endothelial cell proliferation and migration contribute to antiangiogenic effects (9).
Fatigue, nausea, anorexia, and hepatotoxicity with intravenous administration (9)