Make a Contribution to the BTC Today
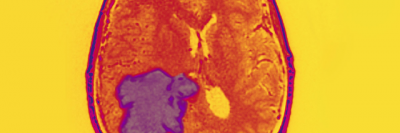
There exists a dire need to improve treatments for malignant brain tumors. Current strategies, including surgery, radiation, and chemotherapies, can have serious side effects and are generally not curative. The Brain Tumor Center (BTC) at Memorial Sloan Kettering Cancer Center provides a unique opportunity to bring together researchers and clinicians for scientific discovery and development of truly effective brain tumor therapies. Together, our physicians and scientists are working to deploy all available modern genomic and transgenic technologies afforded by MSK’s state-of-the-art facilities.
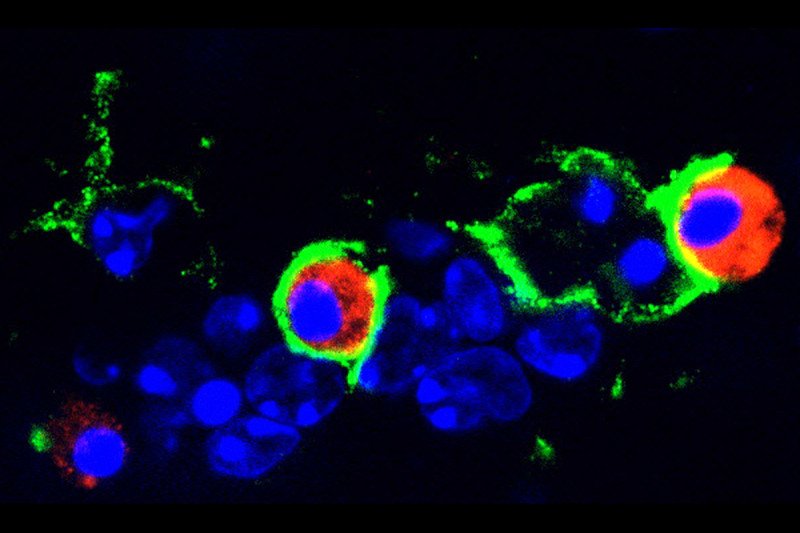
In the face of challenges to more effectively treat glioblastoma (GBM), basic scientific efforts must be supported to better understand the unique underlying biology. The cell or cells that normally give rise to GBM must be identified so that the key changes that take place in transforming them into tumors can be identified.
Modern genomic studies suggest the diversity of GBM subtypes. The functional and clinical significance of these findings must be validated and translated into practical application.
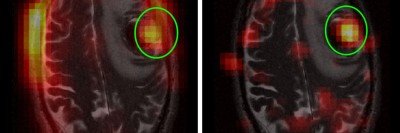
The natural history of tumor development must be better comprehended. The brain microenvironment that likely supports tumor growth must be understood and neutralized. The precise mechanism of tumor growth and development, whether hierarchical or plastic, must be unraveled to establish a baseline for evaluating new therapies.
The BTC is committed to supporting and enhancing collaboration among researchers, as well as the development of tools to advance the fight against brain tumors. Through the establishment of a comprehensive patient-derived xenograft (PDX) program, thoroughly curated GBM samples will be used to develop xenografts for comprehensive molecular profiling. Primary cultures will also be established and genomically mined. Complementary genetically engineered mouse models of GBM will be characterized and maintained.
These long-term resources will be at the disposal of MSK researchers and clinicians for the testing of new hypotheses and for preclinical evaluation.
A seminar series and seed grants for the development of new hypotheses will be supported as a means to maintain a connected research community.
If you would like to obtain further information about the Brain Tumor Center, or donating to the program please contact Michelle Lien by email: [email protected].