Physician-scientist Viviane Tabar
Since the late 1990s, when researchers first reported a technique to isolate and grow human embryonic stem cells, known as ES cells, in culture, experts have deliberated the many possible uses for them. Among these are developing better systems for studying disease processes and creating new cells and tissues to replace those lost to disease or injury.
In a paper published recently in Science, a team from Memorial Sloan Kettering and elsewhere has reported the first-ever genetically engineered model of cancer created from human ES cells. Research enabled by the advance has given hope of a treatment for a rare pediatric brain tumor called diffuse intrinsic pontine glioma (DIPG).
“Recent sequencing studies have provided clues about some of the genetic mutations that characterize these aggressive, incurable tumors, but how the mutations lead to cancer has not been understood,” says physician-scientist Viviane Tabar, the senior author of the study. “We decided to model DIPG using ES cells in an effort to decipher the mechanisms of tumor formation.”
Transforming Stem Cells
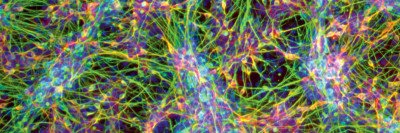
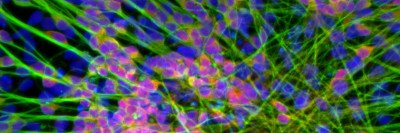
The excitement surrounding human ES cells arises partly from the fact that these cells are pluripotent: They have the ability to develop into almost any cell type in the body. Dr. Tabar is a neurosurgeon, and an area of focus in her laboratory for the past several years has been studying the processes by which ES cells can be induced to form different types of brain cells.
The first step in the current study was transforming ES cells into the type of brain stem cells that are present in young children. The investigators then introduced into those brain stem cells the cancer-causing genes (also called oncogenes) that are expressed in DIPG. When injected into the brain stems of mice, the cells formed tumors that exhibited many of the features of human DIPG.
“On the molecular level, the cells we created resembled brain tumors from patients,” Dr. Tabar explains. “Once they began growing in the mice, we could use them to test drugs that could potentially kill cancer cells in patients.”
A Focus on Epigenetics
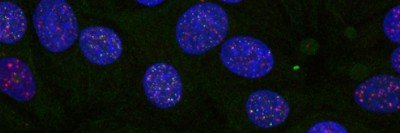
The investigators screened for drugs that could kill DIPG cells while sparing healthy brain stem cells and zeroed in on a single compound, called menin inhibitor-2 (MI-2). Menin is a tumor suppressor — a type of protein that normally prevents cancer growth — and has been linked to several other types of cancer. Specifically, it plays a role in histone modification, a type of epigenetic change that, by adjusting the activity of certain genes, can influence cells’ ability to proliferate and grow out of control.
The investigators tested MI-2 in the mice with DIPG and discovered that it led to significant shrinkage of the brain tumors. They then confirmed that the drug could potentially be effective against the human disease by killing tumor cells that had been removed from patients and grown in a dish in the lab.
“The work shows that an ES-based system for cancer modeling may have significant advantages over tumor cell cultures and earlier types of mouse models,” Dr. Tabar concludes. “In addition, it offers hope for a potential therapy against DIPG, a disease that has proven intractable against hundreds of other drugs and that carries a very short life expectancy.”
David Allis, a Tri-Institutional Professor in MSK’s Molecular Biology Program, a professor at The Rockefeller University, and a leading expert on the study of histones and epigenetic changes, was co-author on the new study. Kosuke Funato, a postdoctoral scientist in Dr. Tabar’s lab, was the first author.