Liao JCF, Gregor P, Wolchok JD, Orlandi F, Craft D, Leung C, Houghton AN, Bergman PJ. 2006. Vaccination with human tyrosinase DNA induces antibody responses that correlate with clinical outcome in dogs with advanced melanoma. Cancer Immunity 6:8
Bergman PJ, Camps-Palau MA, McKnight JA, Leibman NF, Craft DM, Leung C, Liao J, Riviere IK, Sadelain M, Hohenhaus AE, Gregor P, Houghton AN, Perales MA, Wolchok JD. 2006. Development of a xenogeneic DNA vaccine program for canine malignant melanoma at the Animal Medical Center. Vaccine 22:4582-85
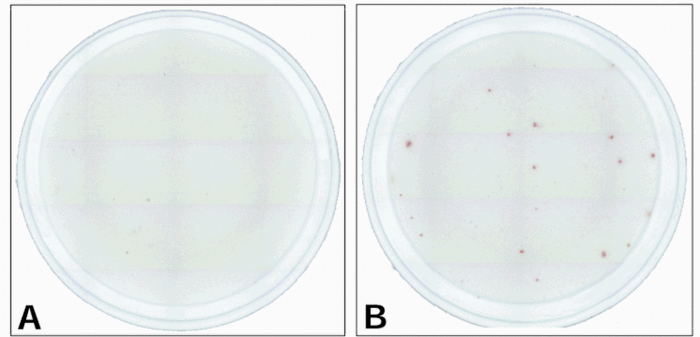
Janetzki S, Schaed S, Blachere NE, Ben-Porat L, Houghton AN, Panageas KS. Evaluation of Elispot assays: influence of method and operator on variability of results.J Immunol Methods. 2004; 291:175-183.
Bergman PJ, McKnight J, Novosad A, Charney S, Farrelly J, Craft D, Sadelain M, Wulderk M, Jeffers Y, Hohenhaus AE, Segal N, Gregor P, Engelhorn M, Riviere I, Houghton AN and Wolchok J. Long term survival of dogs with advanced malignant melanoma following DNA vaccination with xenogenic human tyrosinase: A phase I trial. Clin Cancer Res 2003; 9:1284-1290
Schaed SG, Klimek VM, Panageas KS, Musselli CM, Butterworth L, Hwu W-J, Livingston PO, Williams L, Lewis JJ, Houghton AN, Chapman PB. T cell responses against tyrosinase 368-376(370D) peptide in HLA*A0201+ melanoma patients: randomized trial comparing incomplete Freund’s adjuvant, GM-CSF, and QS-21 as immunological adjuvants. Clin Cancer Res 2002; 20:2610-2615
Lewis JJ, Janetzki S, Schaed S, Panageas KS, Wang S, Williams L, Meyers M, Butterworth L, Livingston PO, Chapman PB, Houghton AN. Evaluation of CD8(+) T-cell frequencies by the Elispot assay in healthy individuals and in patients with metastatic melanoma immunized with tyrosinase peptide. Int J Cancer. 2000;87:391-8.
Janetzki S, Song P, Gupta V, Lewis JJ, Houghton AN. Insect cells as HLA-restricted antigen-presenting cells for the IFN-gamma elispot assay. J Immunol Methods. 2000;234:1-12.
Gilewski T, Adluri S, Ragupathi G, Zhang S, Yao TJ, Panageas K, Moynahan M, Houghton A, Norton L, Livingston PO. Vaccination of high-risk breast cancer patients with mucin-1 (MUCI) keyhole limpet hemocyanin conjugate plus QS-21. Clin Cancer Res. 2000;6:1693-1701.
Janetzki S, Song P, Gupta V, Lewis JJ, Houghton AN. Insect cells as HLA-restricted antigen-presenting cells for the IFN-gamma elispot assay. J Immunol Methods. 2000;234:1-12.
Grant SC, Kris MG, Houghton AN, Chapman PB. Long survival of patients with small cell lung cancer after adjuvant treatment with the anti-idiotypic antibody BEC2 plus Bacillus Calmette-Guérin. Clin Cancer Res. 1999;5:1319-1323.
Nasi ML, Lieberman P, Busam KJ, Prieto V, Panageas KS, Lewis JJ, Houghton AN, Chapman PB. Intradermal injection of granulocyte-macrophage colony-stimulating factor (GM-CSF) in patients with metastatic melanoma recruits dendritic cells. Cytokines Cell Mol Ther 1999; 5:139-144
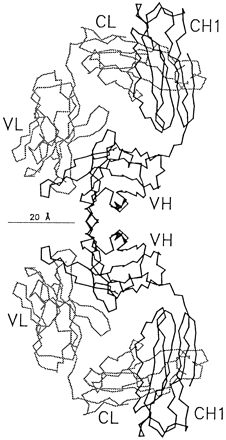
Kaminski MC, MacKenzie CR, Mooibroek MJ, Dahms TES, Hirama T, Houghton AN, Chapman PB, Evans SV. The role of homophilic binding in anti-tumor antibody R24 recognition of molecular surfaces. J Biol Chem 1999; 274:5597-5604
Yao TJ, Meyers M, Livingston PO, Houghton AN, Chapman PB. Immunization of melanoma patients with BEC2-keyhole limpet hemocyanin plus BCG intradermally followed by intravenous booster immunizations with BEC2 to induce anti-GD3 ganglioside antibodies. Clin Cancer Res. 1999;5:77-81.
Nasi ML, Meyers M, Livingston PO, Houghton AN, Chapman PB. Anti-melanoma effects of R24, a monoclonal antibody against GD3 ganglioside. Melanoma Res 1997; 7:S155-S162
McCaffery M, Yao T-J, Williams L, Livingston PO, Houghton AN, Chapman PB. Immunization of melanoma patients with BEC2 anti-idiotypic monoclonal antibody that mimics GD3 ganglioside: enhanced immunogenicity when combined with adjuvant. Clin Cancer Res. 1996;2:679-686.
Jakubowski AN, Bajorin DF, Templeton MA, Chapman PB, Cody BV, Thaler H, Tao Y, Filippa DA, Williams L, Sherman ML, Garnick MB, Houghton AN. Phase I study of continuous-infusion recombinant macrophage colony-stimulating factor in patients with metastatic melanoma. Clin Cancer Res. 1996;2:295-302.
McCaffery M, Yao TJ, Williams L, Livingston PO, Houghton AN, Chapman PB. Immunization of melanoma patients with BEC2 anti-idiotypic monoclonal antibody that mimics GD3 ganglioside): enhanced immunogenicity when combined with adjuvant. Clin Cancer Res. 1996;679-686.
Minasian LM, Yao TJ, Steffens TA, Scheinberg DA, Williams L, Riedel E, Houghton AN, Chapman PB. A phase I study of anti-GD3 ganglioside monoclonal antibody R24 and recombinant human macrophage-colony stimulating factor in patients with metastatic melanoma. Cancer. 1995; 75:2251-2257.
Brooks D, Taylor C, Dos Santos B, Linden H, Houghton A, Hecht TT, Kornfeld S, Taetle R. Phase Ia trial of murine immunoglobin A antitransferrin receptor antibody 42/6. Clin Cancer Res. 1995;1:1259-1265.
Minasian LM, Szatrowski TP, Rosenblum M, Steffens T, Morrison ME, Chapman PB, Williams L, Nathan CF, Houghton AN. Hemorrhagic tumor necrosis during a pilot trial of tumor necrosis factor-alpha and anti-GD3 ganglioside monoclonal antibody in patients with metastatic melanoma. Blood. 1994;83:56-64.
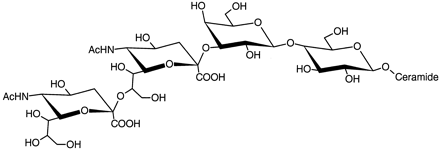
Yuasa H, Scheinberg DA, Houghton AN. Gangliosides of T lymphocytes: evidence for a role in T-cell activation. Tissue Antigens. 1990;35:47-56.
Vadhan-Raj S, Cordon-Cardo C, Carswell E, Minzter D, Dantis L, Duteau C, Templeton MA, Ottegen HF, Old LJ, Houghton AN. Phase I trial of a mouse monoclonal antibody against GD3 ganglioside in patients with melanoma: induction of an inflammatory response at tumor sites. J Clin Onc. 1988;6:1636-1648.
Houghton AN, Mintzer D, Cordon-Cardo C, Welt S, Fliegel B, Vadhan S, Carswell E, Melamed MR, Oettgen HF, Old LJ. Mouse monoclonal IgG3 antibody detecting GD3 ganglioside: a phase I trial in patients with malignant melanoma. Proc Natl Acad Sci USA. 1985;82:1242-1246.