Our laboratory aims at exploiting recent advances in stem cell biology to develop radically new therapies for degenerative disease and cancer. The main focus in the lab is on the biology and application of human embryonic stem (ES) and human induced pluripotent (iPS) cells. Pluripotent stem cells can provide a truly unlimited source for deriving therapeutically relevant cell types for applications in human disease modeling or regenerative medicine.
A major effort of the lab is devoted to harnessing and manipulating the differentiation potential of human pluripotent stem cells. We have developed highly efficient strategies to coax human pluripotent stem cells into specific brain cell populations in a dish. Specific types of nerve cells that are routinely generated in the lab include midbrain dopamine neurons, motoneurons, GABA+ interneurons and sensory neurons.
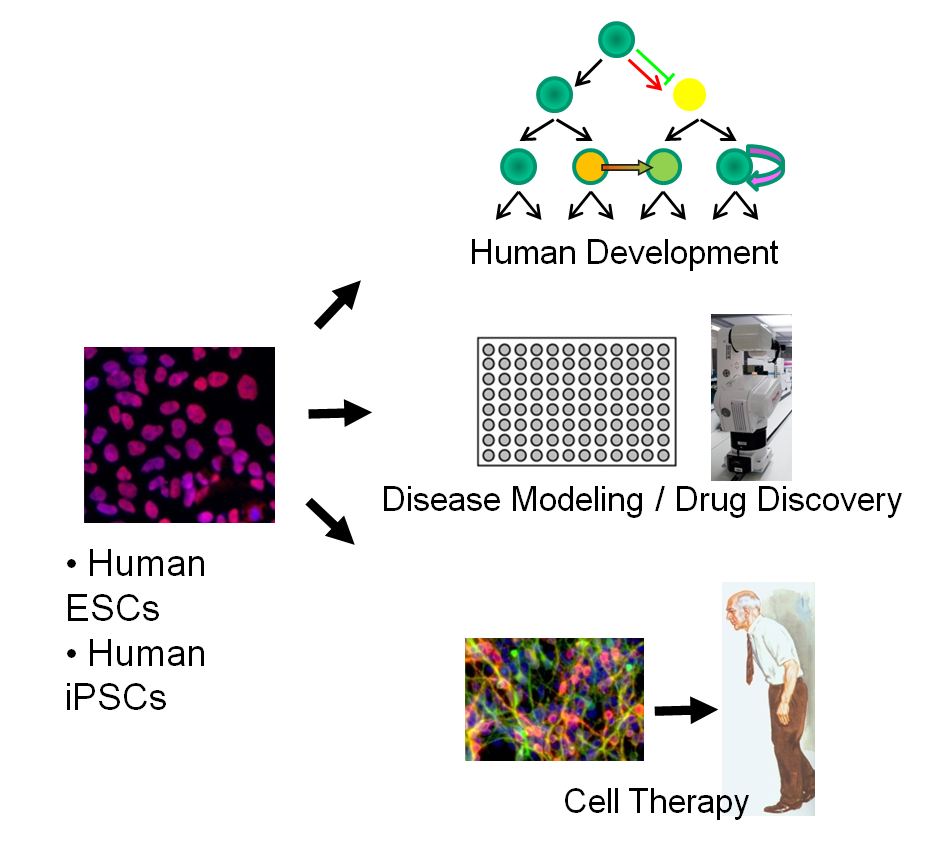
Those neurons are used for modeling or potentially treating human disorders such as Parkinson’s disease, ALS (Lou Gehrig’s disease) as well as neurodevelopmental and psychiatric disease. We are also interested in applications outside the CNS — particularly in musculoskeletal disease and disorders related to cell types derived from the neural crest and sensory placodes. Understanding the molecular signals required for converting stem cells into specialized cell types also offers insights into basic mechanisms of human development. The lab uses both candidate approaches as well high throughput chemical and genetic screens to address such questions in human ES and iPS cells.
Projects in the lab fall into three major categories:
Directing fate and age of human pluripotent stem cells
Our lab has pioneered the directed differentiation of human pluripotent stem cells into multiple specialized cell types of both the central and peripheral nervous system. The long-term goal is to develop a roadmap to re-create the full cellular diversity of the nervous system. In addition to cell fate, we are interested in defining the molecular pathways that control timing of human differentiation including cell fate specification, neuronal maturation and the programming and re-programming of cellular age in a dish.
Modeling human disease using pluripotent stem cells
One of the most intriguing applications of human pluripotent stem cells is the possibility of recreating a disease in a dish and to use such cell-based models for drug discovery. Our lab uses human iPS and ES cells for modeling human disorders of the brain and peripheral nervous system such as Parkinson’s disease, familial dysautonomia, Herpes Simplex encephalitis and melanocyte related disorders including cancer.
Human pluripotent stem cells in cell therapy
We have a long-standing interest in developing stem cell based therapies for regenerative medicine. Our main current effort is on Parkinson’s disease (PD). We have developed protocols to derive midbrain dopamine neurons from human ES and iPS cells capable of engraftment in mouse, rat and monkey models of PD. Based on such data we were awarded a consortium award from NYSTEM to pursue the first clinical application of human ES cell based dopamine neurons in PD patients.
Links to the Labs of Some of Our Recent Collaborators
- Viviane Tabar @ MSKCC
- Mark Tomishima @ MSKCC
- Songhai Shi @ MSKCC
- David Sulzer @ Columbia University
- Jeff Kordower @ Rush University
- Dimitri Krainc @ Northwestern University
- D. James Surmeier @ Northwestern University
- Ole Isacson @ McLean Hospital/Harvard University
- Jean-Laurent Casanova @ Rockefeller University
- Stewart Anderson @ UPenn
- Karl Deisseroth @ Stanford University