Our laboratory uses a combination of cryo-electron microscopy, x-ray crystallography, and functional approaches to study the mechanisms of eukaryotic ion channels involved in calcium signaling and membrane-embedded enzymes.
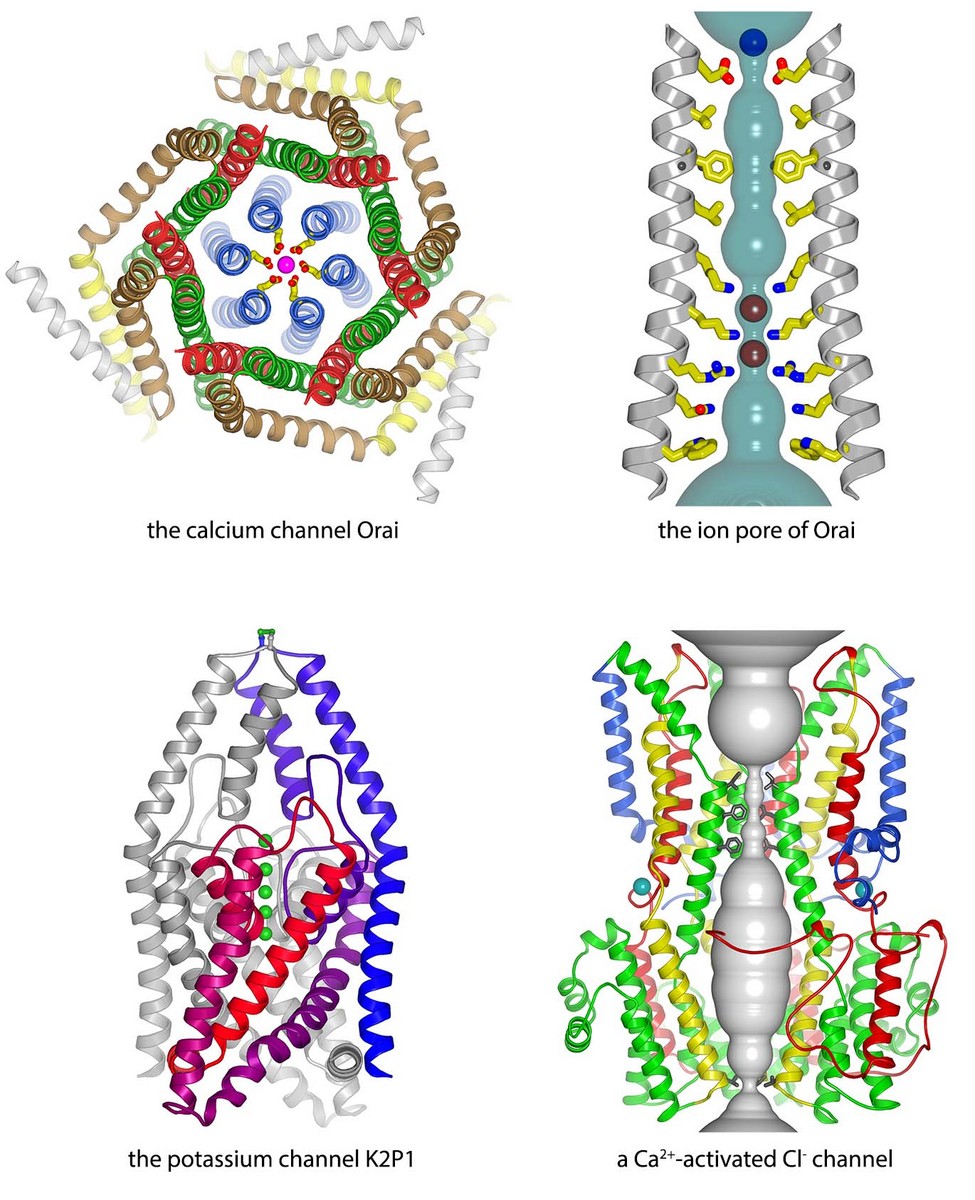
Our primary research focus is to understand the molecular mechanisms of eukaryotic ion channels that generate or respond to intracellular calcium (Ca2+) signals. By obtaining three-dimensional information for transient ion channel complexes complimented by experiments that assay channel function, we aim to discover the mechanisms by which the channels conduct ions across cellular membranes, achieve ion selectivity, and are gated. One effort (e.g. Hou et al. and Hou et al.) is in the study of the Ca2+ release-activated Ca2+ (CRAC) channel Orai, which generates cytosolic Ca2+ signals necessary for activation of immune responses. Through a mechanism that is without precedent, the ion pore of Orai in the plasma membrane is gated by interaction with a protein in the membrane of the endoplasmic reticulum (ER). We also study the mitochondrial calcium uniporter MCU, which is a highly-selective and highly-regulated Ca2+ channel that serves as the major route for Ca2+ uptake by mitochondria (e.g. Baradaran & Wang, et al.). In another effort (e.g. Dickson et al., Vaisey & Long), we are studying a Ca2+-activated chloride channel, BEST1, that is regulated by intracellular Ca2+ signals. Mutations in this channel cause certain eye diseases. In
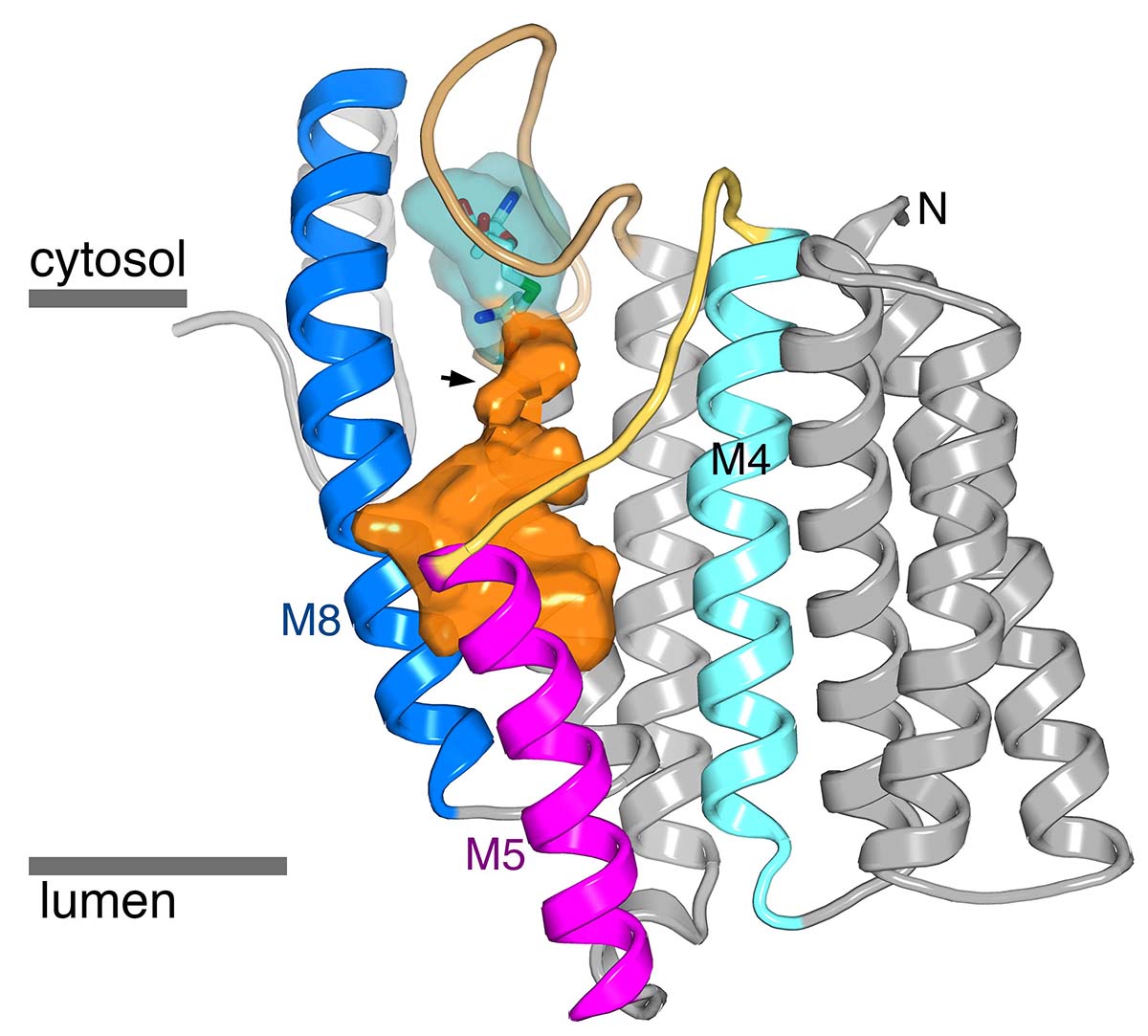
In a second area of research, we aim to address how membrane-embedded enzymes catalyze chemical reactions within lipid membranes. The salient questions include how both water-soluble and lipophilic substrates access the catalytic active site, what conformational changes in the enzymes occur, and what constraints the lipid membrane places on these. Our focus is the enzyme ICMT, which methylates the C-terminus of Ras and other ‘CAAX’ proteins. Our recently-determined structure of ICMT (Diver et al.) reveals that the active site is exposed to both the lipid membrane and the cytosol. The structure informs how the enzyme catalyzes methylation of its diverse substrates. Data suggest that inhibition of ICMT may have utility for combating cancer. Experiments are underway to evaluate inhibitors of ICMT that we developed as anti-cancer agents.
September, 2018 - GSK graduate student successfully George Vaisey defends this thesis! He will soon begin as a postdoctoral fellow in Roderick MacKinnon’s laboratory at Rockefeller University. Congratulations, George!
September, 2018 - Very proud of our first JGP paper! Using electrophysiology and biochemistry, GSK graduate student George Vaisey discovers that the bestrophin channel inactivates and does so through an allosteric mechanism (Vaisey & Long). It’s completely different than the inactivation mechanisms in other channels!
September, 2018 - Xiaowei and Shana’s new paper on the opening of the CRAC channel Orai is published in Elife (Hou, Burstein & Long)!
August, 2018 - Rozbeh and Chongyuan determine structures of the mitochondrial calcium uniporter MCU using cryo-EM (Baradaran, Wang, Siliciano & Long). It’s one cool channel and our first published structure using cryo-EM.
March, 2018 - Xiaowei and Shana’s structure of the calcium channel Orai in an open conformation is online at bioRxiv (Hou, Burstein & Long)
January, 2018 - Melinda’s tour-de-force study describing the structure of ICMT and its implications is out! (Diver et al.)
March, 2017 - Our Titan Krios cryo-EM microscope is fully operational (and just downstairs)!
November, 2016 - GSK Graduate Student George Vaisey publishes a study that identifies regions of the anion channel BEST1 that control ion selectivity and activation. Check it out - it’s a cool channel with surprises! (Vaisey, Miller & Long)
May, 2016 - Steve receives the Boyer Award for achievement in basic research at the Gerstner Sloan Kettering Graduate School commencement.
April, 2016 - BCMB Graduate Student alumna Melinda Diver bids farewell to the Long lab and heads to David Julius’ lab at UCSF for a postdoc! Congratulations Melinda!!
December, 2015 - MSKCC purchases a high-end cryo-EM microscope (FEI Titan Krios with a Gatan K2 Summit detector) that will support ongoing research in the Long laboratory and other laboratories at MSKCC.
Summer, 2015 - Steve and postdocs Alex Miller and Xiaowei Hou are visiting scientists at the Janelia Research Campus of HHMI in Niko Grigorieff’s laboratory to learn cryo-EM.
Summer, 2015 - Lab Alumna, Veronica Kane Dickson takes a position at the Cambridge Institute for Medical Research to begin her own lab in 2016! Congratulations!
August, 2015 - George Vaisey receives a PhD Fellowship from the Boehringer Ingelhein Fonds. George is in the GSK graduate program.
November, 2014 - Popstdoc Xiaowei Hou is selected among all postdocs in the New York area as a Blavatnik Regional Award Finalist for her pioneering work in the structural elucidation of the calcium release-activated channel (CRAC) Orai - see the announcement and video here.
October, 2014 – Congratulations to Veronica and Leanne for their work on the structure and function of a eukaryotic calcium-activated chloride channel (Abstract).
September, 2014 – BCMB Graduate Student Melinda Diver receives her PhD!
July, 2014 – Congratulations to Melinda for her recent publication on ICMT (Abstract).
May, 2014 – Xiaowei Hou receives the MSKCC Postdoctoral Researcher Award at the Gerstner Sloan Kettering Graduate School commencement.
If you are interested in joining the laboratory, please email Stephen Long. Postdoc/Staff Scientist applicants with broad expertise in electrophysiology are encouraged to apply. Prospective graduate students are encouraged to apply to the Gerstner Sloan Kettering Graduate School of Biomedical Sciences, the Weill Cornell Graduate School of Medical Sciences, and/or the Tri-Institutional Training Program in Chemical Biology (TPCB).