The intestinal epithelium, composed of intestinal epithelial cells (IECs) connected by tight junctions, creates a physical and biochemical barrier to separate luminal organisms from intestinal tissues. The intestinal mucosa is in contact with harmless commensal bacteria as well as potential pathogens, thereby functioning to immunologically survey the intestinal lumen.
IECs monitor the tone of the lumen, recognizing a variety of microbial products in both antigen-independent and antigen-dependent manners to participate in coordinated immune tolerance or, alternatively, immune response. Interestingly, many aspects of intestinal immune homeostasis fail to develop in the absence of the intestinal microbiota. Germ-free mice show extensive defects and impaired development of gut-associated lymphoid tissues and antibody production. Adding to the complexity of microbiota composition are host and environmental factors, which can modulate microbiota composition and include age, antimicrobial use, disease, inflammation, metabolites, stress, and diet, particularly malnourishment.
A role for the microbiota has been examined in relation to a variety of clinical outcomes, ranging from obesity and atherosclerosis, to allergies and asthma, and even to cancer development and autism. Hematopoietic stem cell transplantation (HSCT) has been no exception, with several groups recently uncovering important relationships between the microbiota and outcomes in HSCT recipients.
In a paper by Robert Jenq et al. we demonstrated that in murine and human recipients of HSCT, intestinal inflammation secondary to GVHD is associated with major shifts in the composition of the intestinal microbiota, and, in turn, gut flora can modulate the severity of intestinal inflammation. Mice that developed GVHD exhibited a reduction in flora diversity with an expansion in the order of Lactobacillales and decrease in the order of Clostridiales. This change in microbiota diversity was also observed in human settings in patients with GVHD.
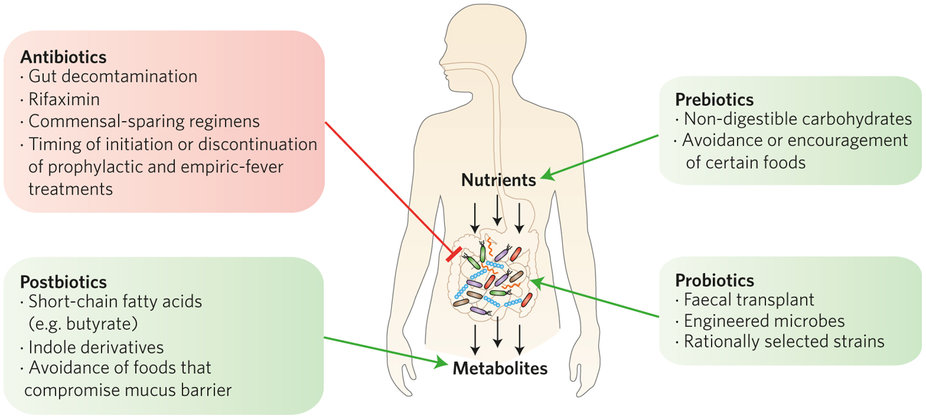
Patients receiving an allogeneic hematopoietic stem cell transplant often develop neutropenic fever, which is treated with antibiotics. Such antibiotic treatment also may inadvertently wipe out beneficial intestinal bacteria that reduce gut inflammation. The work by Yusuke Shono et al. demonstrated that transplant patients treated with certain broad-spectrum antibiotics show an increase in graft-versus-host disease in the colon. Analysis of a mouse model revealed that this was due to loss of beneficial gut bacteria and overgrowth of bacterial strains that consumed the protective mucus layer of the colon rendering it more susceptible to inflammation and injury.
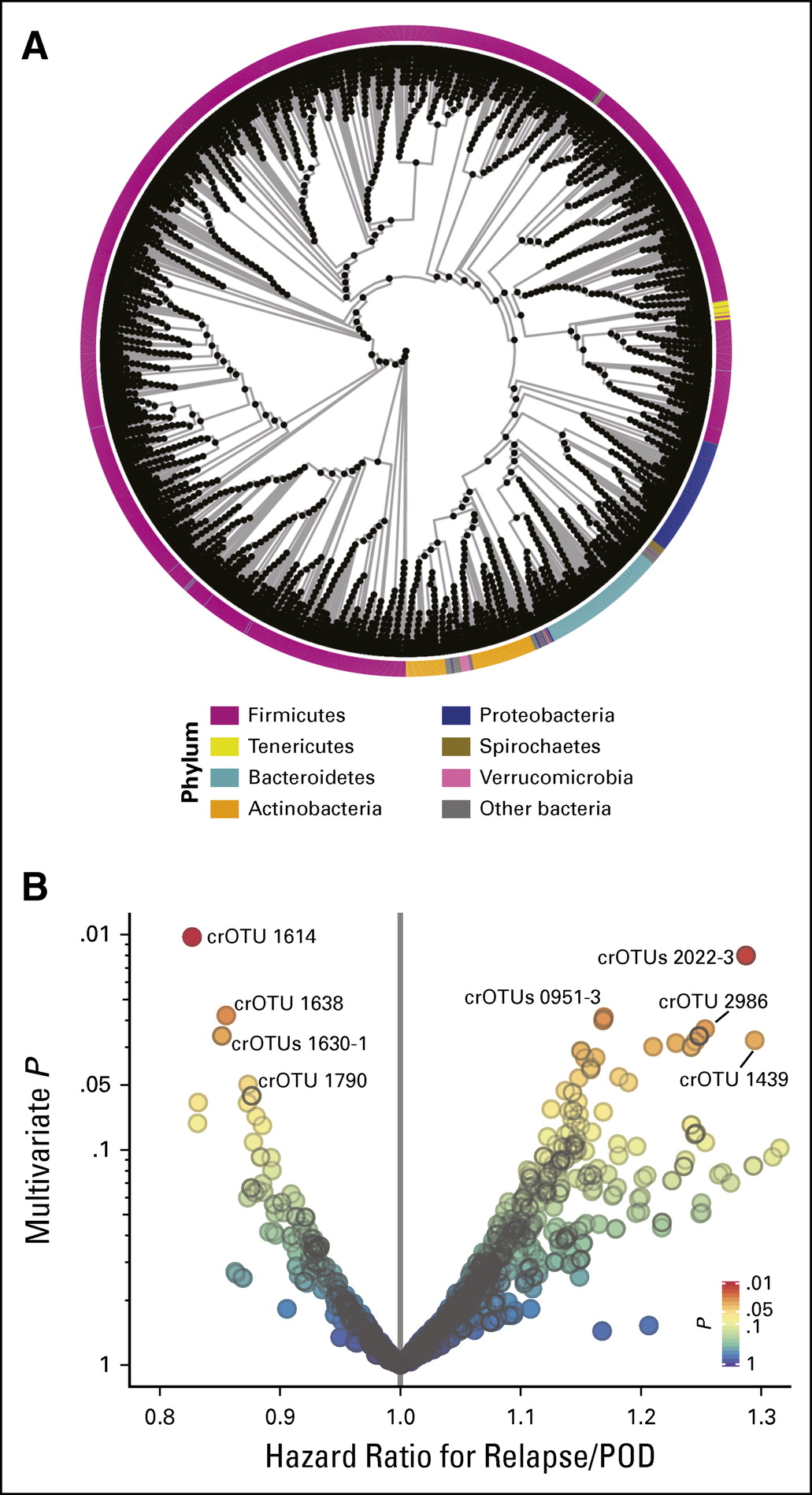
The healthy bacteria in our guts have an important influence on many aspects of health. In this study of over 500 MSKCC patients, a certain group of gut bacteria were found to predict whether relapse of leukemia would occur after bone marrow transplantation. This group of bacteria, whose presence or abundance predicts less relapse and improved survival after allo-HSCT, includes the commensal organism Eubacterium limosum and several other related species. This paves the way for future studies that will optimize intestinal bacteria to improve patient health.