Understanding adaptive responses to stressors and how these differ between normal and diseased tissues remains unsatisfactorily addressed. A key to addressing this unresolved biological question is to study how stressors impact tissue-specific ’interactomes’, the intricate proteomewide cellular networks of proteins linked through interactions. In this context interactomes are maps of how stressors, including genetic lesions, proteotoxic and environmental insults, individually or combined, alter protein-protein interaction (PPI) networks and perturb the system as a whole.
Our program takes advantage of properties of protein-protein interaction networks (i.e. interactome networks) to understand, diagnose and treat diseases, such as cancer and neurodegenerative diseases.
It aims to investigate the identity and the architecture of interactome networks in cells exposed to chronic molecular and environmental stressors with the goal of understanding disease mechanisms and identifying vulnerabilities.
It aims to take advantage of these vulnerabilities to discover and develop drug candidates, biomarkers, diagnostics and treatment strategies.
Our ultimate goal is the translation of our discoveries from bench to bedside.
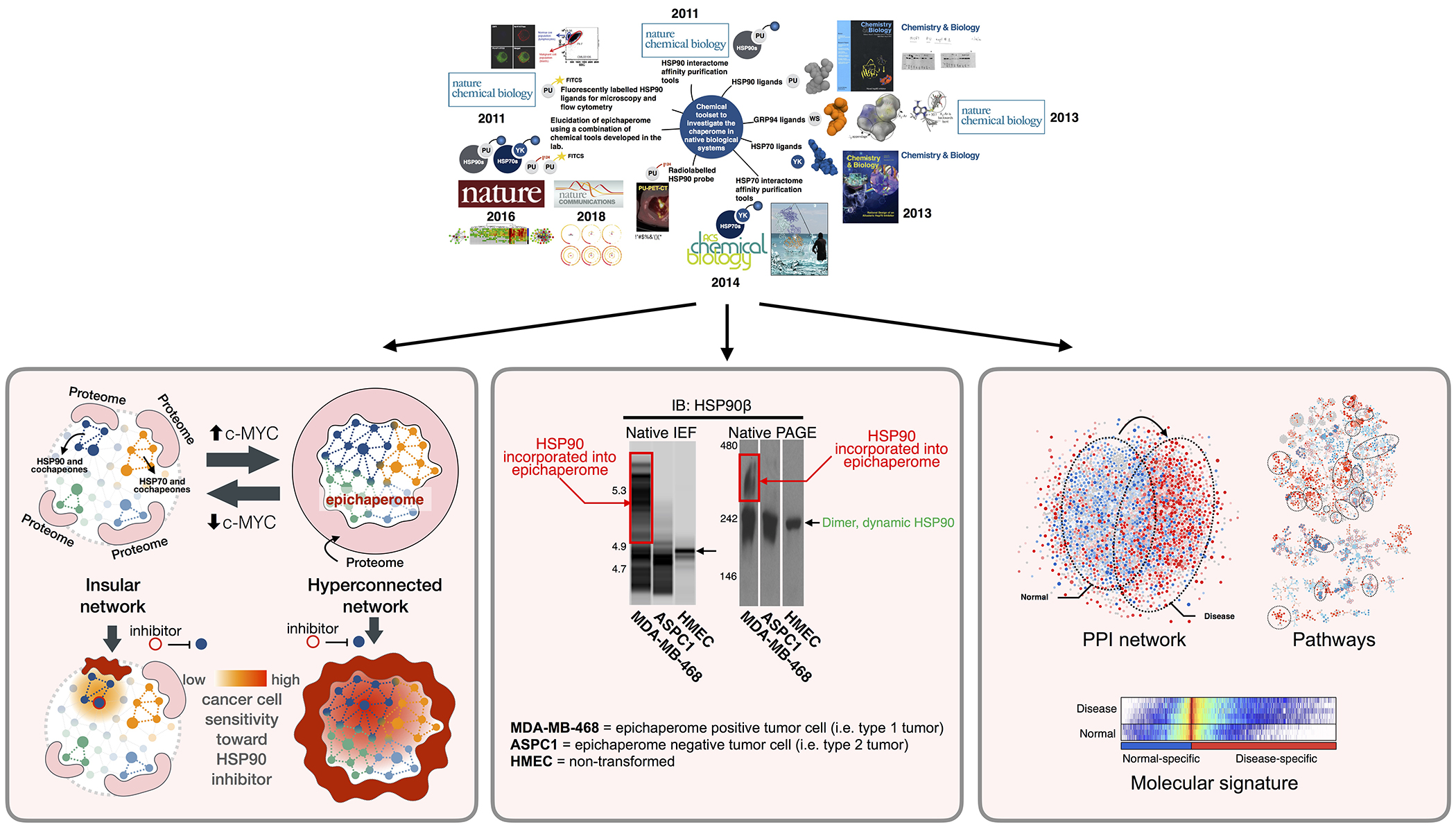
Rodina A, Xu C, Digwal CS, Joshi S, Patel Y, Santhaseela AR, Bay S, Merugu S, Alam A, Yan P, Yang C, Roychowdhury T, Panchal P, Shrestha L, Kang Y, Sharma S, Almodovar J, Corben A, Alpaugh ML, Modi S, Guzman ML, Fei T, Taldone T, Ginsberg SD, Erdjument-Bromage H, Neubert TA, Manova-Todorova K, Tsou MB, Young JC, Wang T, Chiosis G. Systems-level analyses of protein-protein interaction network dysfunctions via epichaperomics identify cancer-specific mechanisms of stress adaptation. Nat Commun. 2023;14(1):3742. doi: 10.1038/s41467-023-39241-7. https://www.nature.com/articles/s41467-023-39241-7
Ginsberg SD, Sharma S, Norton L, Chiosis G. Targeting stressor-induced dysfunctions in protein-protein interaction networks via epichaperomes. Trends Pharmacol Sci. 2023;44(1):20-33. doi: 10.1016/j.tips.2022.10.006. https://pubmed.ncbi.nlm.nih.gov/36414432/
- Featured in: Alzforum, National Institute on Aging Featured Research, Neurology Today Editor’s Pick, ClinicalOMICs, Must read on https://flipboard.com/topic/tau, https://www.usagainstalzheimers.org/news/january-17-2020 and others Altmetric 116.
Pillarsetty N, Jhaveri K, Taldone T, Caldas-Lopes E, Punzalan B, Joshi S, Bolaender A, Uddin MM, Rodina A, Yan P, Ku A, Ku T, Shah SK, Lyashchenko S, Burnazi E, Wang T, Lecomte N, Janjigian Y, Younes A, Batlevi CW, Guzman ML, Roboz GJ, Koziorowski J, Zanzonico P, Alpaugh ML, Corben A, Modi S, Norton L, Larson SM, Lewis JS, Chiosis G*, Gerecitano JF, Dunphy MPS. (*lead contact) Paradigms for Precision Medicine in Epichaperome Cancer Therapy. Cancer Cell. 2019 Nov 11;36(5):559-573.e7. doi: 10.1016/j.ccell.2019.09.007. Epub 2019 Oct 24.
- Cover of Cancer Cell November 11, 2019; Highlighted in Cancer Cell November 11, 2019; Featured on StressMarq blog November 27, 2019; MSKCC blog, https://www.mskcc.org/blog/bull-s-eye-imaging-technology-could-confirm-…. Altmetric 59
Featured in:
- Kai Bartkowiak & Klaus Pantel. Cancer: A shocking protein complex. Nature (2016), News&Views, doi:10.1038/nature19476
- Stu Borman. Heat-shock protein complexes serve as cancer drug targets. ‘Epichaperomes’ that form in some cancer cells might enable selective therapies. CE&N News, Volume 94 Issue 40, pp. 10-11 Science & Technology Concentrates; October 10, 2016
- Andrew P. Han. Assays for Heat Shock Protein Complexes Could Serve as Companion Dx for Inhibitor Drugs. GenomeWeb, Proteomics & Protein Research Oct 10, 2016
- Anna Dart. Networking: a survival guide. Nature Reviews Cancer | Published online 11 Nov 2016; doi:10.1038/nrc.2016.125
- Heidi A. Dahlmann. Chaperome Complexes Influence Tumor Survival. ACS Chem. Biol., 2016, 11 (11), pp 2941–2943. DOI: 10.1021/acschembio.6b00969
- Philipp Markolin. How a complex network of proteins contributes to cancer survival. https://medium.com/advances-in-biological-science/how-a-complex-network…
- https://f1000.com/prime/726802982
- https://www.mskcc.org/blog/experimental-cancer-drug-developed-msk-leads…
- http://www.samustherapeutics.com/
- https://www.prnewswire.com/news-releases/samus-therapeutics-announces-l…
- Highlighted, MSKCC blog https://www.mskcc.org/blog/experimental-cancer-drug-developed-msk-leads…, Featured in Alzforum, Faculty1000, etc. Altmetric 40




