Another major focus of my laboratory is to translate the mechanistic findings and the chemical biology tools into treatment strategies. These efforts encompass a large spectrum of activities such as lead compound discovery and development, medicinal chemistry, computational analyses, assay development, pharmacokinetic and pharmacodynamic studies, in vivo efficacy studies, biomarker discovery, diagnostic assay development, combination therapy studies, intellectual property development, and commercialization strategies.
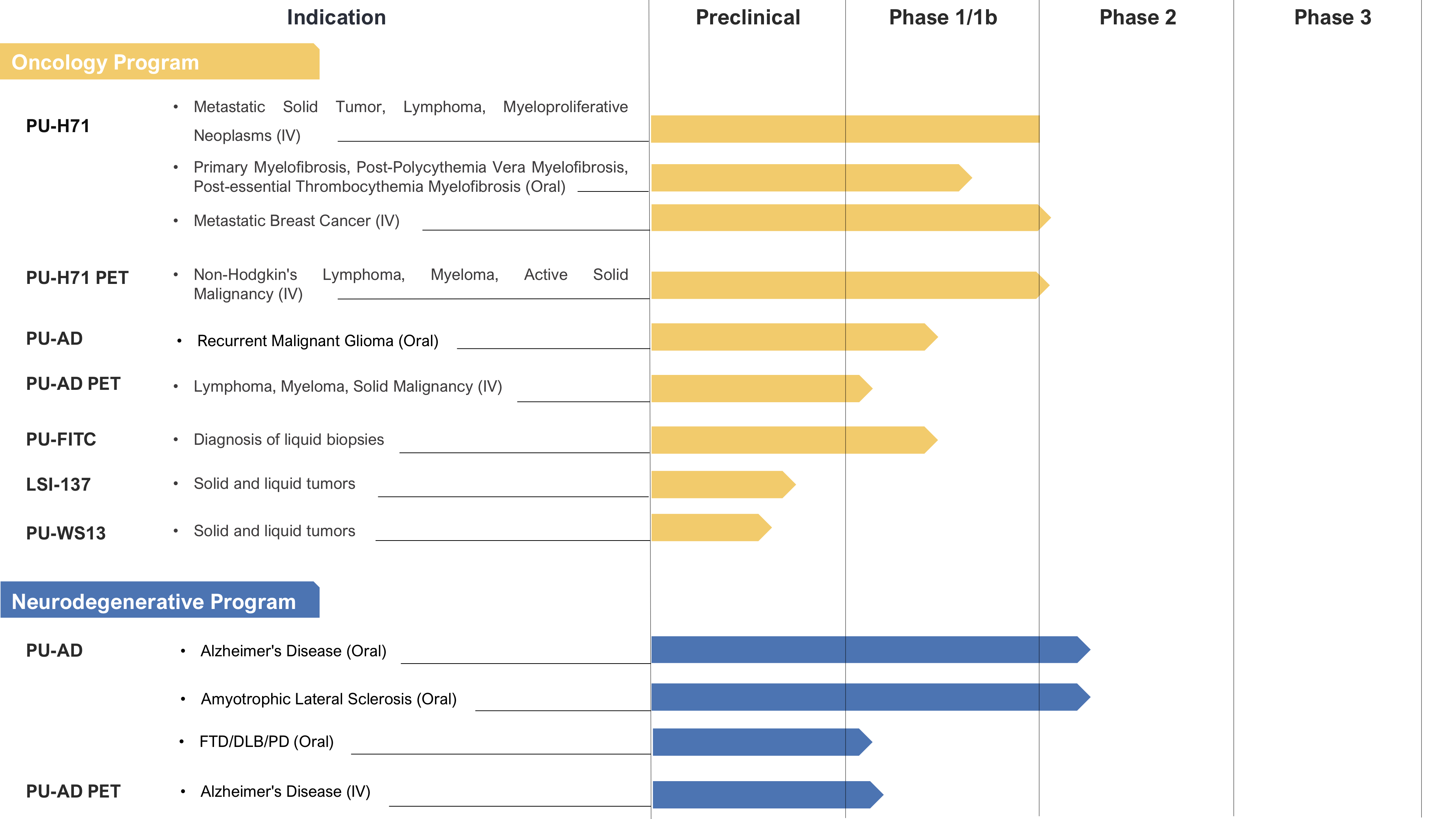
We also use the chemical tools to tailor them into companion diagnostics and potential drugs for cancer and other diseases such as in neurodegeneration. No invention becomes a drug or a diagnostic without strong intellectual property and a commercialization plan behind it; therefore, my lab is directly involved in writing and maintaining the over 550 patent applications and granted patents covering our portfolio and is behind the creation of start-ups to develop the inventions toward the clinic and the market. So far, we have translated to clinic the PU-H71 inhibitor and its companion diagnostics PU-PET and PU-FITC; this agent, together with the companion diagnostics is now in Phase 1b/2 clinical studies (Cancer Cell 2019, JCO Precision Oncology 2020, npj Precision Oncology 2021). To advance this portfolio, we have formed a startup, Samus Therapeutics Inc. Recently we have translated to clinic the CNS directed diagnostic 124I-PU-AD PET, and the therapeutic, PU-AD, for the treatment of Alzheimer’s disease (now in Phase 2 studies, Nature Comm 2020), the therapeutic Icapamespib in glioblastoma (Phase 1b, Nature Comm 2021) and are moving forward alternative approaches of targeting the epichaperomes (via targeting Glyc62GRP94 and via allosteric HSP70 targeting).
Chiosis lab receives no industrial funding or support.
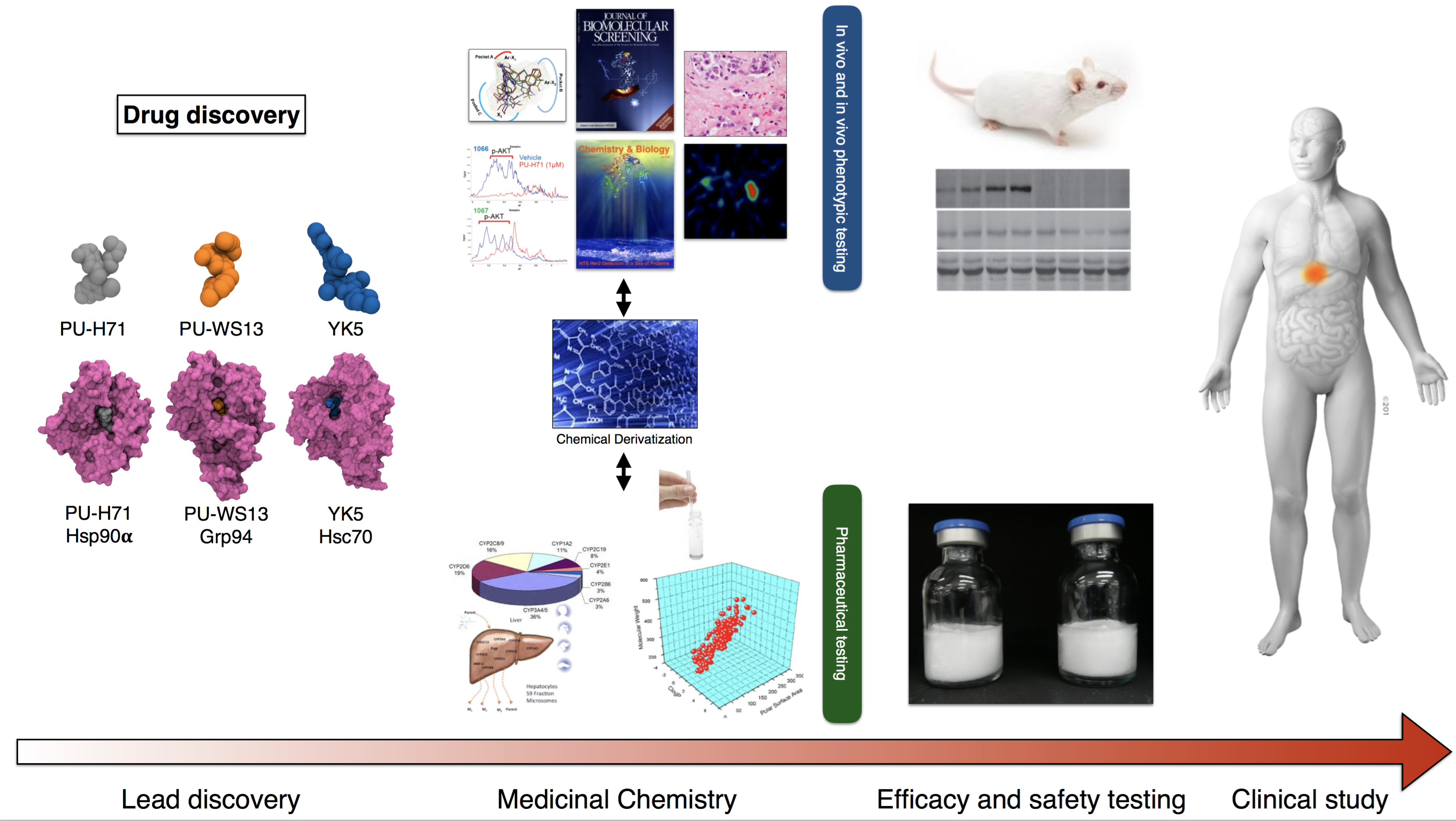

Dr. Tony Taldone from my lab shares his thoughts on the challenges and rewards of drug discovery: