Success rates of standard treatments for inoperable primary pancreas and liver tumors are poor, but a radiotherapy technique that combines classical, modern, and novel concepts is offering renewed hope of long-term survival for these patients.
Stereotactic ablative radiation therapy (SABR) or hypofractionated ablative radiotherapy incorporates image guidance and internal organ motion control to deliver radiation doses high enough to reliably ablate tumors.
Ablation of the Tumor Is Key
Radiation therapy is used as a standard curative treatment option in many different solid tumors. SABR is a recent radiotherapy innovation that has been a successful technique to ablate small lung tumors that are not near critical organs. Another treatment that is commonly used is low-dose stereotactic body radiotherapy (SBRT). It uses the same stereotactic delivery techniques that are used with the SABR method but must be delivered at reduced doses in order to protect sensitive organs such as those near the pancreas and liver. Patients and physicians often confuse the SABR with SBRT. Unfortunately, this reduced dose does not deliver the necessary ablative effect (1).
The reduced-dose approach of SBRT came about because initial studies of its use at higher doses in tumors near the gastrointestinal tract reported significant toxicity to surrounding organs. To counteract this side effect, physicians lowered the doses to 25-33Gy, given over three to five days, for liver and pancreatic tumors to ensure the treatment is well tolerated. This is roughly half the dose needed for ablation of even small tumors.
Refining Stereotactic Ablative Radiation Therapy
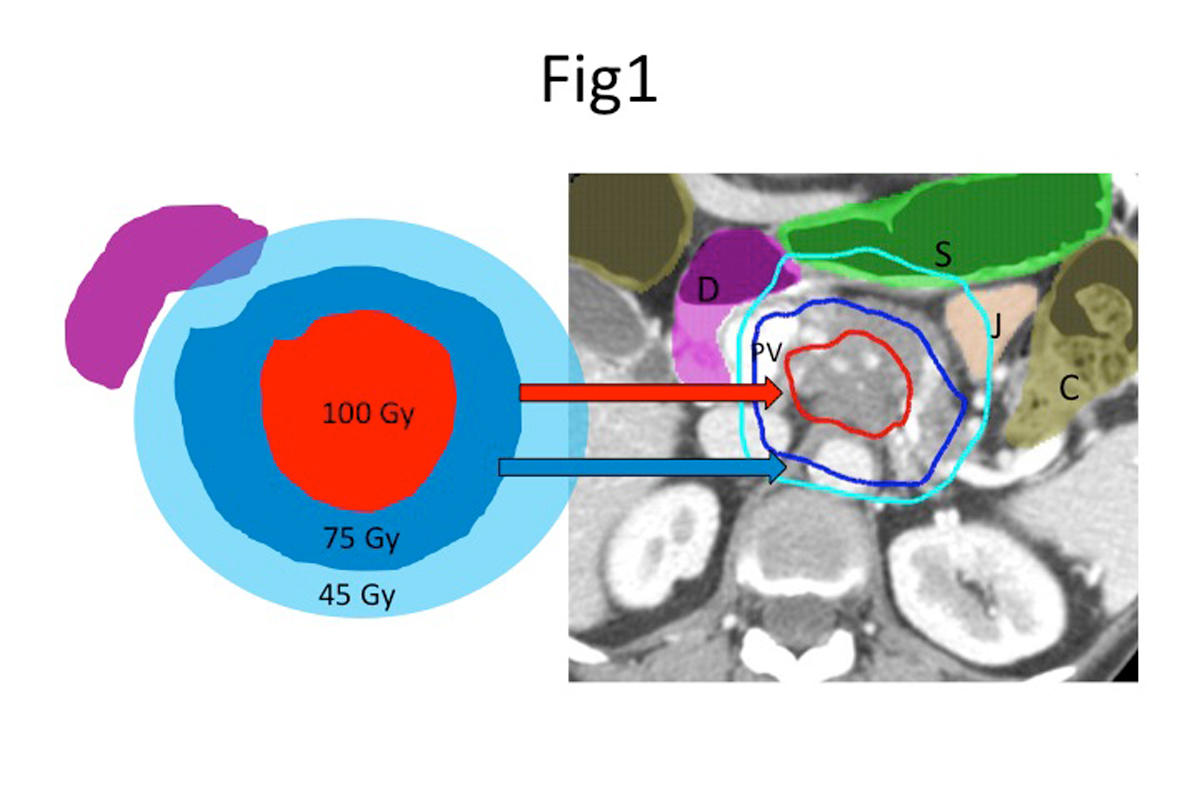
The emergence of SABR and its success in lung cancer led to its use in ablating liver metastases. SABR (50Gy delivered in five days) achieves excellent results (>90% local tumor control at five years) for small lung and liver tumors that are not amenable to resection or other ablative approaches. These experiences led to the exploration of the use of SABR for larger, inoperable primary pancreatic and liver tumors.
Over the past decade, my colleagues and I have used several strategies to overcome the issue of toxicity risk and subsequent damage to critical organs such as the stomach and duodenum. By increasing the number of treatments to allow time for repair of radiation damage to normal tissues between treatments, incorporating the use of daily CT image guidance, and employing meticulous internal organ motion control, we have made it possible to deliver ablative doses safely.
At Memorial Sloan Kettering, our current approach is to deliver 75Gy in 25 fractions or 67.5Gy in 15 fractions (Figure 1) (3), which are both equivalent to the effective ablative dose given in one week (50Gy in 5 days), and twice as high as the lower dose of SBRT that is given commonly for pancreatic and liver cancers.
Internal organ motion control is one of the novel aspects of SABR and is achieved by patients practicing a breath-holding technique while they undergo treatment. Tracking, abdominal compression, and gating are all options for respiratory motion control that are used to reduce dose to normal tissue and escalate dose to tumors. At MSK we use a technique of feedback-guided inspiratory breath-hold gating using the Varian Real-Time Position Management system.
Results of Stereotactic Ablative Radiotherapy Over Three to Five Weeks for Pancreas Tumors
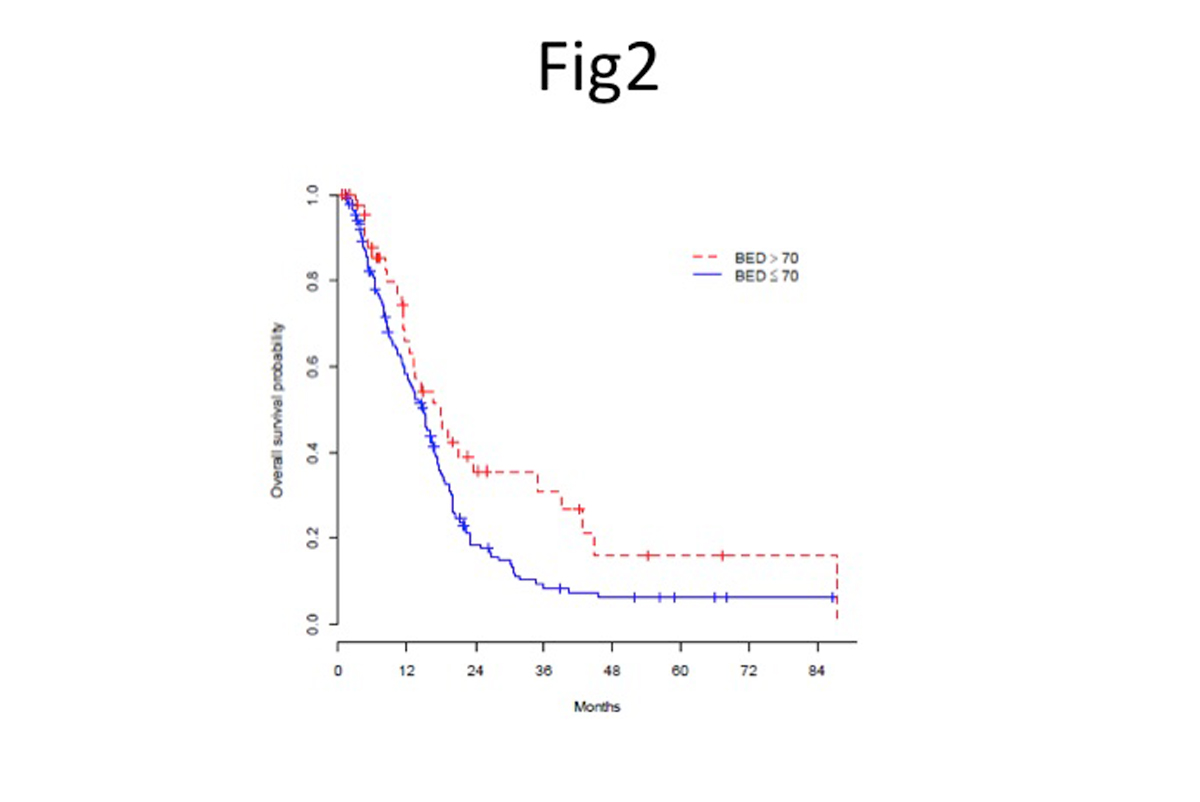
Local tumor progression leads to mortality in up to half of all patients with locally advanced pancreatic cancer. My colleagues and I have recently published results of a study demonstrating that ablative doses of radiation can lead to long-term survival in such patients. According to our data, 35% of patients with unresected tumors who are alive at three years received an ablative dose. The longest survivors are alive after more than eight years (Figure 2)(1),(2).
Results of Stereotactic Ablative Radiotherapy (SABR) Over Three to Five Weeks for Liver Primary Tumors
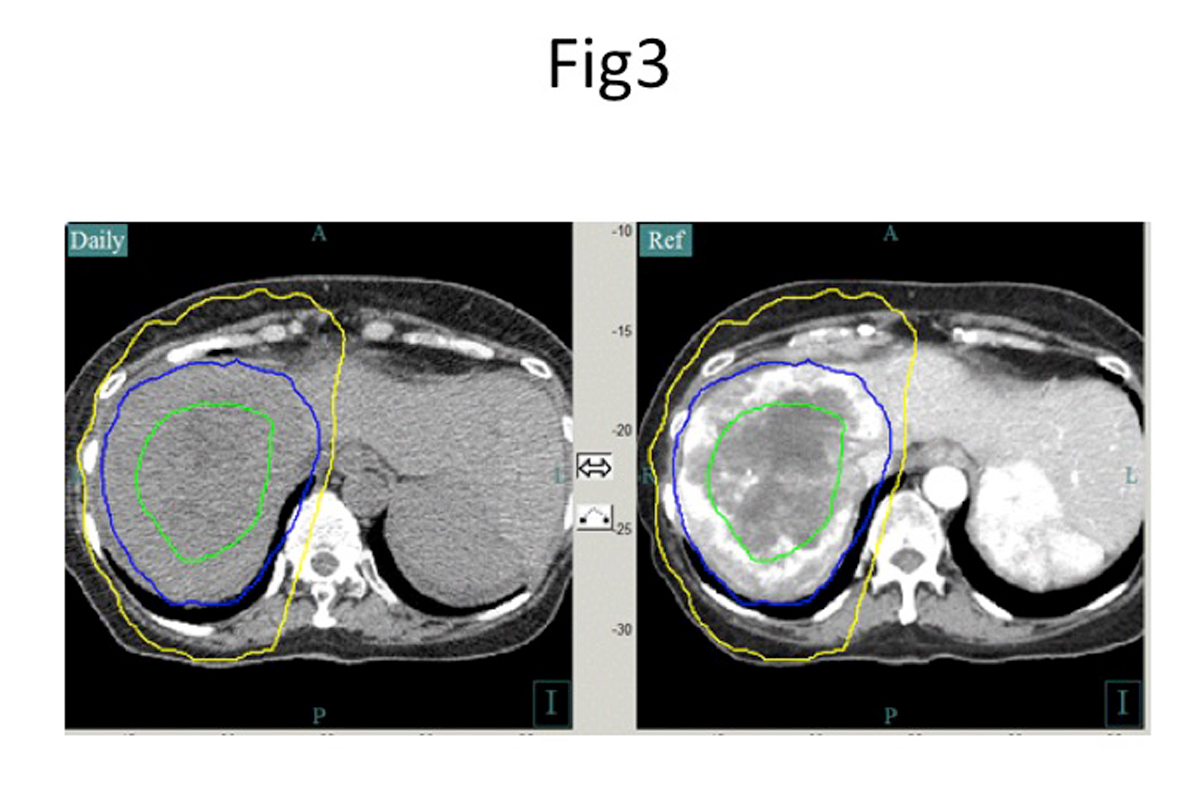
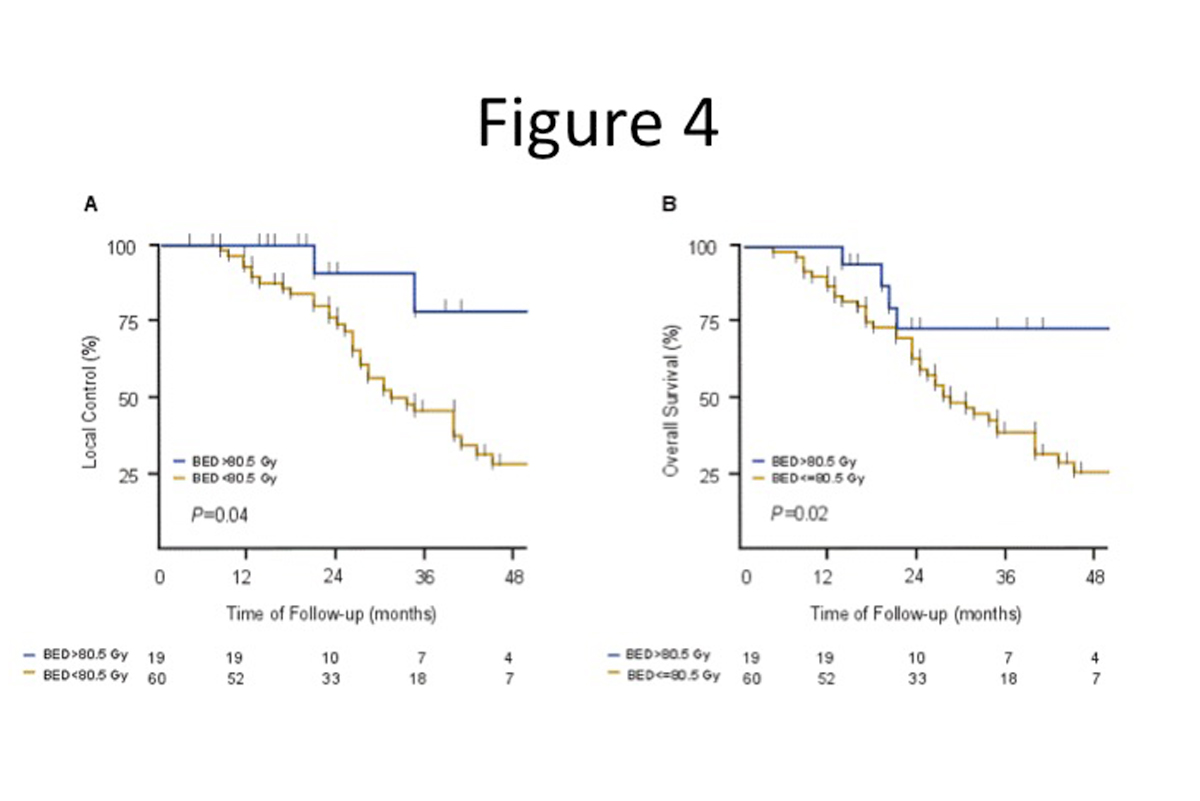
For much the same reasons as pancreatic cancers, the sensitivity of nearby organs such as the bile duct makes large liver tumors challenging to treat with an ablative dose. Additionally, the presence of underlying liver disease, the sensitivity of the liver parenchyma to radiation, and internal organ motion due to respiration and luminal filling make treatment difficult. We address these challenges by using the same methods of controlling respiratory motion and CT image guidance to assess the position of the bowel (Figure 3) and by increasing the number of treatments. Adopting this approach has resulted in high long-term local tumor control and survival rates in inoperable hepatocellular carcinoma (90%) (4) and intrahepatic cholangiocarcinoma (IHC) (80%)(4),(5). Most of the IHC patients had initially received chemotherapy and were then selected for the use of radiotherapy. Effective local tumor control correlated with the use of an ablative dose, prevented tumor-related liver failure, and led to a substantial survival benefit (Figure 4). Failure to control the local tumor resulted in tumor related hepatic failure in 89% of patients.
In summary, we have refined a technique over the last decade that allows ablative doses of radiation to be delivered with low risk of toxicity to tumors near critical organs such as the stomach and duodenum. This approach offers hope to patients with inoperable primary pancreas and liver and tumors that long-term survival is possible.
- Crane CH: Improving Long-Term Survival in Patients With Locally Advanced Pancreatic Cancer via the Delivery of Definitive Radiotherapy Doses. Oncology (Williston Park) 29:561-2, 566, 2015
- Crane CH: Hypofractionated ablative radiotherapy for locally advanced pancreatic cancer. J Radiat Res, 2016
- Krishnan S, Chadha A, Suh Y, et al: Focal dose escalation for locally advanced pancreatic cancer patients receiving consolidative chemoradiation therapy after induction chemotherapy improves overall survival. Int J Radiat Oncol Biol Phys Under review, 2015
- Hong TS, Wo JY, Yeap BY, et al: Multi-Institutional Phase II Study of High-Dose Hypofractionated Proton Beam Therapy in Patients With Localized, Unresectable Hepatocellular Carcinoma and Intrahepatic Cholangiocarcinoma. J Clin Oncol 34:460-8, 2016
- Tao R, Krishnan S, Bhosale PR, et al: Ablative Radiotherapy Doses Lead to a Substantial Prolongation of Survival in Patients With Inoperable Intrahepatic Cholangiocarcinoma: A Retrospective Dose Response Analysis. J Clin Oncol 34:219-26, 2016
