Lead Contacts
Robert Downey, MD, FACS
Thoracic Service, Department of Surgery
Memorial Sloan Kettering Cancer Center
Takeshi Irie, MD, PhD
Department of Anesthesiology and Critical Care Medicine
Memorial Sloan Kettering Cancer Center
Sam Hellman, PhD
Medical Physics
Memorial Sloan Kettering Cancer Center
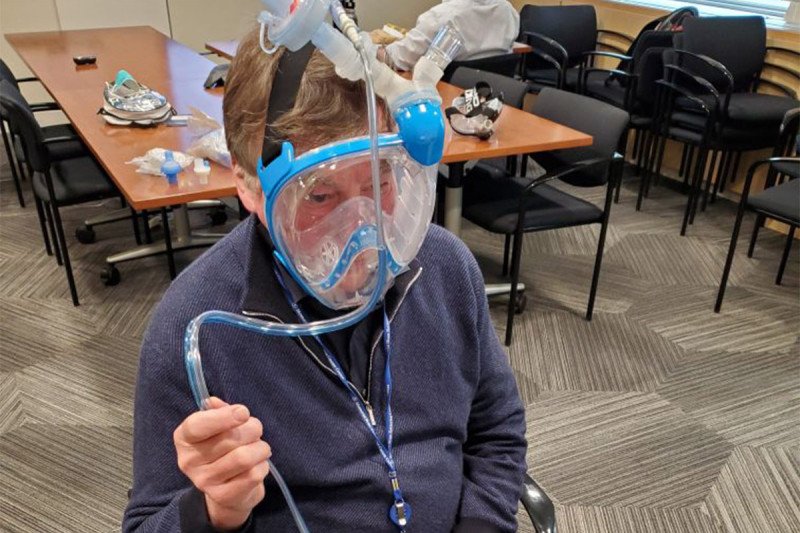
A multidisciplinary team at Memorial Sloan Kettering has modified a commercially available snorkeling mask to address the current shortage of standard clinical respiratory protective equipment.
The simple modifications include a high-grade air filter used in anesthetic circuits and a 3-D printed adapter to connect the mask to the filter.
While it has not yet been used clinically during patient care, the device has performed as designed at the level of an N100 protective device during standard respiratory fit testing.
Built and tested over three weeks in late April 2020, the mask can be constructed easily and quickly to facilitate rapid, widespread adoption while providing basic functionality and reliability.
The team, consisting of biomedical and device-development engineers, surgeons, and anesthesiologists, envisages an application in the global health system in emergency scenarios and in areas with chronic shortages of such protective equipment.
Why a snorkel mask rather than a scuba mask?
A full-face mask allows protection of the mouth, nose, and eyes. A full-face snorkel mask costs one tenth the cost of a full-face scuba mask (approximately $80 versus $800 respectively).
What was the mask type chosen?
Design requirements
The quality of commercially available snorkeling masks varies considerably. A high-quality snorkeling mask has the following characteristics:
- No mixing of inhaled and exhaled air, as rebreathing carbon dioxide (CO2) could potentially cause elevated levels in blood, provoking user discomfort, or theoretically in severe cases, altered mental status from CO2 narcosis
- Silicon is preferred over PVC for a better seal (PVC, being less flexible, does not conform to facial contours as well)
- Minimize “dead space”; dead space is the air column that moves into and out of a person’s respiratory system without reaching the lung alveolae, where the exchange of oxygen and CO2 occurs.
- This video discusses snorkel mask design very clearly.
Commercially available mask chosen: Cressi®
Cressi is a reputable and readily available Italian brand of scuba and snorkeling equipment recommended by local diving professionals. The Cressi Duke dry full face mask meets the criteria above and costs approximately $70 retail. Less expensive masks ($30 per mask) are available, but there are concerns that these are of inferior design for this purpose, which may lead to mixing of inhaled and exhaled air, causing CO2 retention by the user.
One limitation of commercially available snorkeling masks is that glasses cannot be worn with them because the ear pieces of the glasses extend through the seal between the mask and the surface of the face, leading to leakage. One commercial solution, which we have not explored, is available from Ocean Reef®.
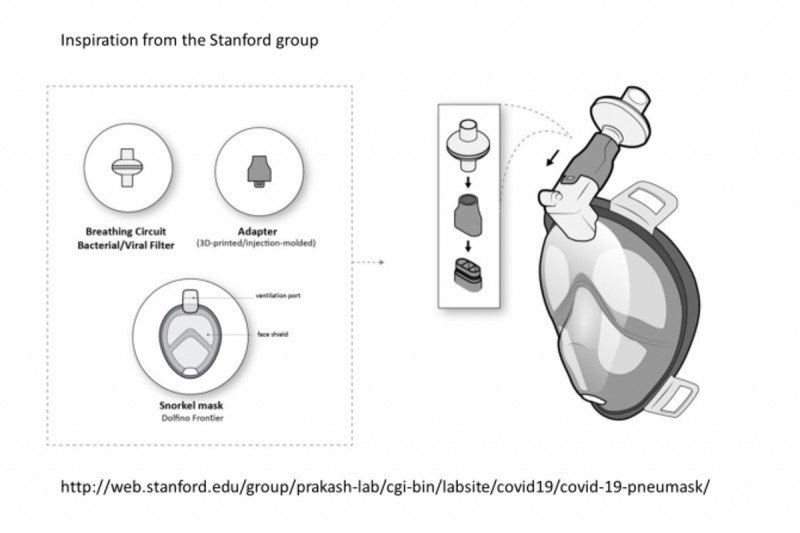
How was the adapter developed?
The snorkel of the mask is already removable for ease of transport. We proposed substituting an N100-quality air filter for the snorkel. However, the orifice on the snorkel is square, while the orifice of the filter is round, necessitating an adapter to attach the mask to the filter. A 3-D printed adapter was created.
Measurements were made with calipers, and it was fit tested by printing small test print rings first. The whole adapter was designed once the base fit was verified.
Materials used for the 3-D adapter
- We first used Stratasys® MED610, which is tested and certified for biocompatibility (skin and mucosal contact and breathable gas) and sterilizable, but it proved to be very brittle. It was necessary to print a thicker adapter to prevent cracking and other problems.
- Markforged® Nylon was found to be more durable.
- PLA (multiple brands) is inexpensive and easy to print.
Shape
Our initial design maintained the mask design of separate inlets and outlets to keep inhaled and exhaled air separate. However, experimentation suggested that this was causing increased effort to breathe, while a single inlet-outlet adapter did not lead to symptomatic CO2 retention during short periods of mask use and did lead to reduced effort to breathe. It is important to note that we have not yet tested for CO2 retention.
How were the filters chosen?
Readily available, standard gas filters used in anesthetic circuits were obtained. We used the Dräger SafeStar® 55.
Iterative Improvements as a Result of Serial Fit Testing
Fit Testing
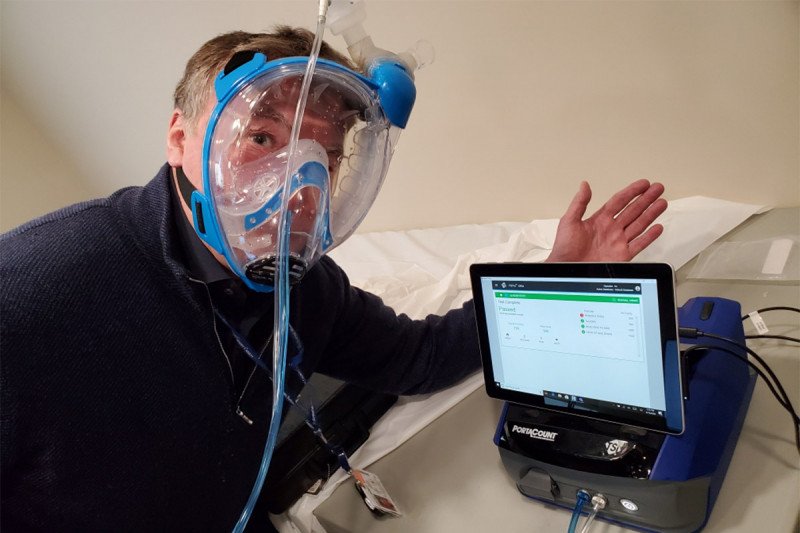
A fit test evaluates the quality of the seal between the respirator’s facepiece and the user’s face. There are two types of fit tests: qualitative and quantitative. A qualitative fit test is a lower-level test that employs a user’s sense of taste and smell to assess the degree of filtration provided without providing a quantitative measure of the amount of leakage. Basically, a substance that we can taste or smell (such as saccharin) is placed outside of the mask and the user reports if they can taste or smell it. Quantitative fit testing measures the actual amount of leakage around the mask into the face. We performed quantitative fit testing using a TSI Portacount® Respirator Fit Tester 8038 device to evaluate the effectiveness of the mask-adapter-filter combination.
Face shape and mask size
Cressi® Duke masks are made in two sized (small-medium and medium-large). Our three test subjects were adult men. Two used the medium-large and one used a small-medium deliberately to test the tolerance of the fit (even though he would likely be more appropriately be fitted with a medium-large).
What were the results with the penultimate and final prototypes?
Three adult men underwent fit testing four times (one was tested twice 24 hours apart). One test was performed with the first version of the adapter, which maintained separation of inhaled and exhaled gases. The second to fourth sessions were performed with the adapter that combines inhaled and exhaled air.
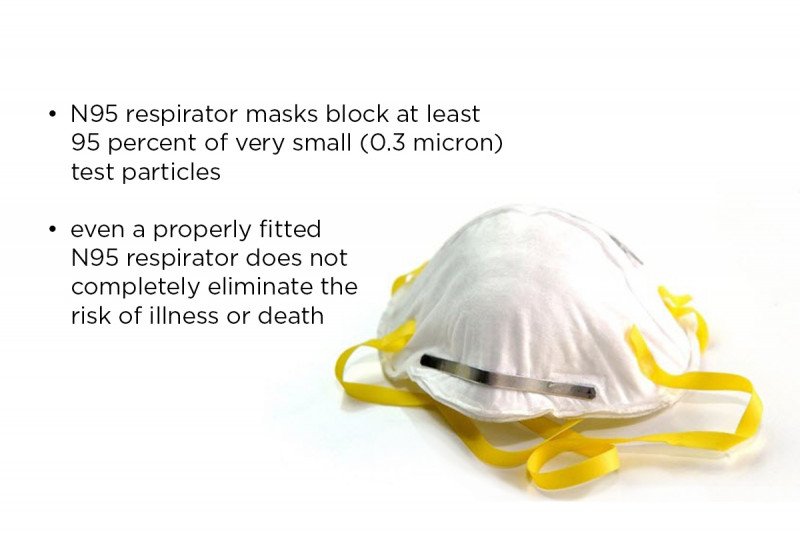
All four sessions were rated as pass, which means that the device exceeded the level of filtration of an N95 mask (i.e., a fit factor of 500 or filtered more than 95% of particles). The final version of the adapted mask has a fit factor of 3176, which means that all but 1 out of 3,176 particles (99.97% of particles) were filtered.
What other tests could be run, and which are necessary?
Tests not felt necessary to perform:
- Qualitative fit testing — We did not perform this, as it is a lower level of testing than a quantitative fit test, which we did perform.
Further tests reasonable to consider performing:
- Carbon dioxide level test — In every person, a certain amount of inhaled air does not participate in the exchange of oxygen and CO2 as it remains in the trachea and bronchi. This amount of an inhaled breath is called the dead space. A snorkel and a mask increase the dead space, leading to decreased expiration of CO2 from the lungs. The degree to which a circuit leads to decreased CO2 expiration can be tested; we did not perform this testing.
- Exercise test — Exercise increases CO2 production. A system that provides adequate clearance of CO2 in a resting user may not provide adequate clearance during exercise. We did not test CO2 clearance during increased levels of activity.
-
Clinical usability decontamination procedures — Given the expense and availability of the mask, a multiple-use design, rather than a single-use one is preferred. Decontamination steps would be needed between uses. Two concerns about decontamination are that the procedure could lead to the degradation of the device’s functionality (e.g., clouding of the faceplate, limiting visualization, and a decreased seal with the face, limiting the effectiveness of isolation from ambient air). Multiple methods of decontamination are available; we have not tested any of these:
- Autoclaving
- Bleach
- Ethanol
- Dry heat at 65 degrees C
- Boiling water
- Feasibility of use — Visual acuity is an important part of many clinical procedures. The faceplate of the mask distorts vision. The degree to which this distortion impairs the performance of procedures has not been tested. Of most concern would be impairment of head and neck surgical procedures and oral surgery procedures.
- Durability of protection — The stability of the passing score has not yet been evaluated for duration of prolonged wear.
Summary
Construction of a high-level respiratory device from readily available materials (a commercially available snorkeling mask and anesthesia circuit filters, along with a 3-D printed adapter) is feasible.
This device has not been tested clinically for ease of use (i.e., comfort during extended use), durability, feasibility (e.g., acuity of vision through the snorkel mask), or effectiveness of decontamination.
This device has not been approved for personnel protection by the US Food and Drug Administration or the National Institute for Occupational Safety and Health.
Contributors
Paul Booth, MS, Biomedical Engineering, MSK
Grant Chen, MD, Department of Anesthesiology and Critical Care Medicine, MSK
Robert Downey, MD, FACS, Thoracic Service, Department of Surgery, MSK
Samuel Hellman, PhD, Medical Physics, MSK
Takeshi Irie, MD, PhD, Department of Anesthesiology and Critical Care Medicine, MSK
Todd Liu, MD, Department of Anesthesiology and Critical Care Medicine, MSK
Ouathek Ouerfelli, PhD, Organic Synthesis Core Facility, Sloan Kettering Institute
Stanford University – Manu Prakash lab
Oceanreefgroup (commercial) (have solution for eye glasses)
Waag: Dutch initiative combining Decathlon mask with P3 filter to a PPE mask (in Dutch only)
Initiative of dutch asbestos industry to produce high graded PPE
Air-Wave (combines snorkel with positive flow of air)
CustomSurgical (open-source instructions) (includes link to 3-D adapter model for Cressi)
Ocean Reef Group (commercially available adapters to allow attachment of filters to snorkeling mask)