All radiotherapy treatments for prostate cancer should be accompanied by image-guidance of the prostate position to rule out significant setup errors in fractional delivery.
The results of our most recent analysis of planning data for 102 patients treated consecutively at Memorial Sloan Kettering indicate that improvements in PSA outcomes with image-guided intensity-modulated radiotherapy (IG-IMRT) and intensity-modulated radiotherapy (IMRT) without any image guidance, were likely due to increased dosimetry for extracapsular disease. (1)
Although dosimetry values for IG-IMRT compared to IMRT without image guidance were similar, computer simulations of set up errors revealed uncertainty in planned doses external to the prostate when image guidance was not employed, tracing to positioning uncertainties. (1)
At MSK we pioneered the use of image-guided techniques to ensure maximum precision to within a millimeter when delivering radiation to prostate cancer tumors. Our most recent study is an example of our continuous efforts to improve hypofractionated radiotherapy to increase survival outcomes and ensure low rates of incontinence and rectal problems in patients with prostate cancer.
MSK Precise™ is our most advanced treatment program for prostate cancer to date. It comprises hypofractionated radiotherapy, real-time image-guidance, MRI planning, and the use of a hydrogel spacer between the rectum and the prostate. Together, these advances are poised to help more men with prostate cancer achieve the best possible outcomes, while minimizing the adverse effects of treatment.
Hypofractionated Radiotherapy
Hypofractionated radiotherapy is a form of external beam radiation that uses advanced imaging and computing technologies to deliver very high doses of radiation with an accuracy of under a millimeter. One of the advantages of this technology is that patients only require five treatment sessions over the course of a little over a week, compared to 45 to 48 sessions delivered over ten weeks with the conventional approach. Further, each treatment with MSK Precise takes three to four minutes, significantly faster than other forms of stereotactic radiosurgery, such as CyberKnife®, which takes nearly an hour per session.
Experts at Memorial Sloan Kettering Cancer Center have been pioneering the use of MSK Precise for over a decade. (2) As of November 2018, we have treated over 1000 patients. Results to date indicate that a majority of patients tolerate MSK Precise well, and less than two percent of patients experienced significant urinary or rectal problems as a result of the treatment. Notably, a substantial percentage of our patients treated with 40 Gy in five fractions have had negative prostate biopsies two years post-treatment.
At the recent ASTRO conference in October 2018, a multi-institutional consortium presented findings from the largest analysis to date examining long-term outcomes with stereotactic radiotherapy. Results showed that stereotactic radiotherapy was safe and effective for the treatment of low- and intermediate-risk prostate cancer in a cohort of 2,142 men enrolled in Phase II trials from 2000 to 2012. At seven years, 95.5 percent of patients in the low-risk group, 91.4 percent of patients in the favorable intermediate-risk group, and 85.1 percent of the unfavorable intermediate-risk group were biochemical-recurrence free. Overall survival rates for the low-risk, favorable intermediate-risk, and unfavorable intermediate-risk groups were 91.4, 93.7, and 86.5 percent respectively. Only 100 patients (6 percent) experienced recurrences and only ten patients (0.6 percent) developed distant metastases. (3)
Advanced Imaging Techniques
At MSK, we pioneered the use of image-guided techniques to improve the accuracy of radiotherapy for prostate cancer treatment. In addition to spearheading IG-MRT, MSK is the only center in the world to routinely use MRI mapping to provide superior visualization of the prostate and surrounding normal tissues. Our team of radiation oncologists collaborates with the medical physics team to ensure we target the prostate with the utmost accuracy during the three to four minutes of actual treatment.
During treatment, we place three small gold markers into the prostate. The markers allow us to precisely locate the prostate in real time, ensuring accurate delivery of high doses of radiation to tumor tissue while minimizing the amount of normal tissue included in the target margin.
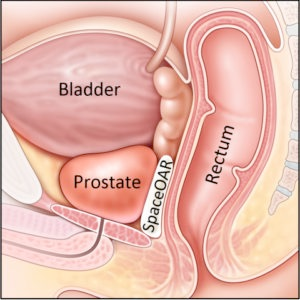
Hydrogel Spacer
Another essential component of the MSK Precise program is the use of an absorbable gel “spacer” that is inserted between the prostate and the rectum. It provides a buffer between the prostate and the rectum, pushing the rectum out of the way of high radiation doses to reduce the risk of bowel side effects.
A prospective Phase III clinical trial conducted by Hamstra et al. found that the hydrogel spacer was safe, well tolerated, and resulted in a significant dose reduction to the rectum. The benefits of the hydrogel spacer at 15 months for quality of life and bowel toxicity were maintained or had increased at the three-year median follow-up. The study examined results for 222 men from 20 participating institutions receiving radiotherapy for prostate cancer, randomized to receive a spacer or no spacer. (4)
In a recent study at MSK, we examined an expanded use of this hydrogel spacer to the low-dose-rate (LDR) brachytherapy setting. Our results showed that it effectively reduced radiotherapy doses to the rectum with minimal toxicity, without affecting prostate and urethral dosimetries. (5)
Advancing Prostate Cancer Care
In total, with MSK Precise we are combining the most recent advances to ensure the best possible outcomes for patients receiving radiotherapy while minimizing the burden of treatment.
Our Prostate Cancer Team of specialists in medical oncology, radiation oncology, urology, urology surgery, pathology, and radiology is recognized worldwide for diagnosing and treating prostate tumors. We continue to look for ways to improve oncologic outcomes, safety and effectiveness of treatments, and improving the quality of life for patients post-treatment. We are currently conducting 40 clinical trials investigating advances in radiotherapy, chemotherapy, immunotherapy and combination therapies.
- Munck Af Rosenschold P, Zelefsky MJ, Apte AP, et al. Image-guided radiotherapy reduces the risk of under-dosing high-risk prostate cancer extra-capsular disease and improves biochemical control. Radiat Oncol. 2018;(13)1:64.
- ClinicalTrials.gov. Dose Escalation Study Using Ultra-Hypofractionated, Image-Guided, Intensity-Modulated Radiotherapy in Prostate Cancer. https://clinicaltrials.gov/ct2/show/NCT00911118
- American Society for Radiation Oncology (ASTRO) Annual Meeting. Men with low- and intermediate-risk prostate cancer can safely benefit from fewer, higher-dose radiation treatments. News Release: October 22, 2018.
- Hamstra DA, Mariados N, Sylvester J, et al. Continued Benefit to Rectal Separation for Prostate Radiation Therapy: Final Results of a Phase III Trial. Radiat Oncol. 2017;97(5):976–985.
- Taggar AS, Charas T, Cohen GN, et al. Placement of an absorbable rectal hydrogel spacer in patients undergoing low-dose-rate brachytherapy with palladium-103. Brachytherapy. 2018;17(2): 251–258.