Recently, significant advances in our understanding of the tumor microenvironment and how tumors interact with the immune system have led to the development of two exciting therapeutic approaches for gynecologic cancers: immune checkpoint inhibitors and armored CAR T cells.
Immune Checkpoint Inhibitors
Novel agents called checkpoint inhibitors work by activating the immune system to attack tumors and deactivate the pathways that make tumors resistant to the immune system. Two examples are programmed death ligand-1 (PD-L1)/PD-1 and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4). See Figure 1.
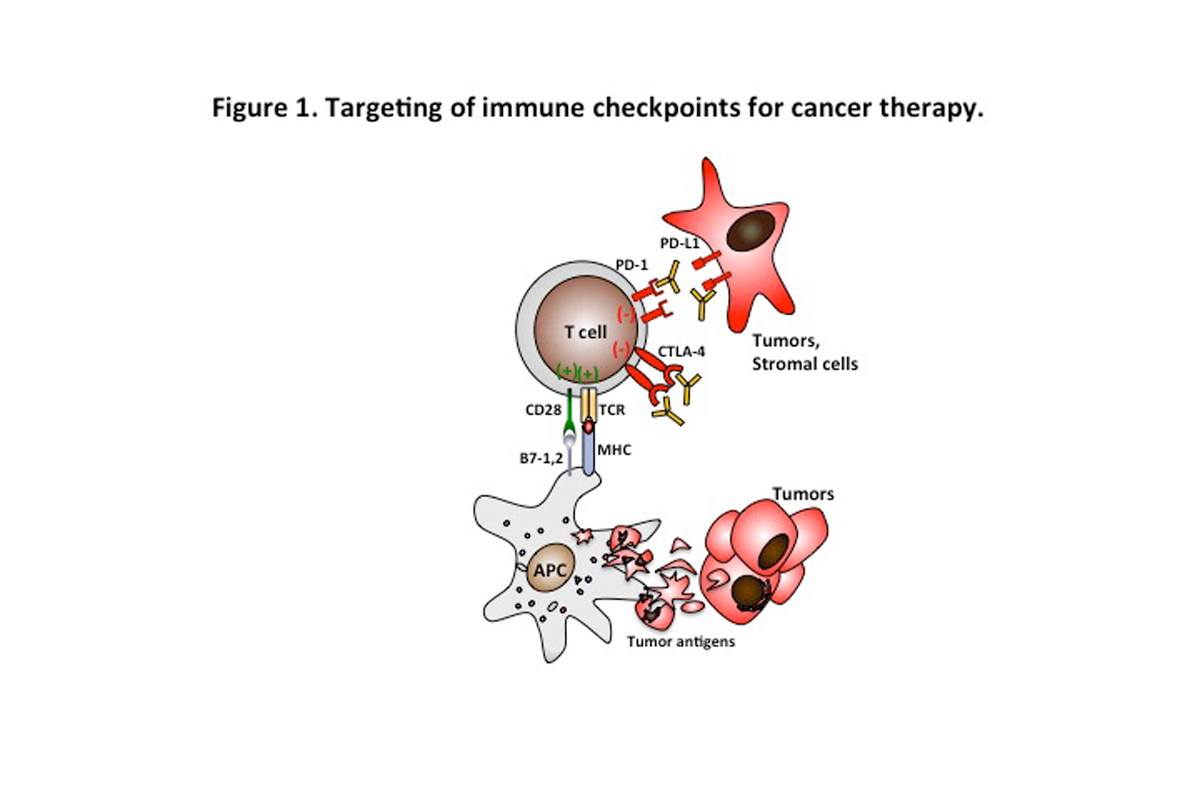
This PD-1/PD-L1 immune checkpoint blockade has already demonstrated benefit in various other types of cancer, including melanoma and lung, bladder, and kidney cancers. Combination treatment with PD-1 and CTLA-4 has shown longer progression-free survival compared with monotherapy. This finding led to FDA approval of the combination treatment of nivolumab (anti-PD-1 antibody) plus ipilimumab (anti-CTLA-4 antibody) for patients with unresectable or metastatic melanoma. (1)
With ample evidence showing the immune system likely plays a key role in eliminating and controlling the growth of gynecologic malignancies, immune checkpoint inhibitors are now being studied in these cancers as well. (2) The NRG Oncology GY002 phase II study (NCT02257528) is evaluating the efficacy of nivolumab in patients with cervical cancer, and the NRG Oncology GY003 randomized phase II study (NCT02498600) is comparing the combination of nivolumab plus ipilimumab with nivolumab alone in patients with relapsed platinum-resistant ovarian cancer. Memorial Sloan Kettering Cancer Center (MSK) is a lead accrual site for these national trials, which have completed first-stage accrual and are awaiting interim analyses.
Despite the success of the immune checkpoint blocking antibodies, the clinical benefit appears to be limited to a subset of patients. This calls for the identification of biomarkers to predict treatment response and for the development of new treatment combinations. To address this issue, all ongoing trials include a range of correlative studies, in order to help predict responders and to delineate the mechanisms of resistance that could help guide future combination therapies. (3) Several ongoing and upcoming trials at MSK are evaluating PD-1/PD-L1 blocking antibodies in combination with other agents. Two trials are currently recruiting patients with recurrent ovarian cancer: a phase I/II trial of the anti-PD-L1 antibody durvalumab in combination with Toll-like receptor 8 agonist motolimod and pegylated liposomal doxorubicin (NCT02431559); and a phase I/II study of the colony stimulating factor 1 receptor inhibitor PLX3397 in combination with the anti-PD-1 antibody pembrolizumab (NCT02452424). In another trial, we are investigating whether the addition of anti-PD-1 therapy can augment the immune response induced against a WT1 peptide vaccine in patients with recurrent ovarian cancer who are in a second or further remission. We are also conducting a phase II study of the multikinase inhibitor lenvatinib in combination with pembrolizumab in patients with recurrent endometrial cancer (NCT02501096). Finally, we are testing combinations of immune checkpoint inhibitors with drugs targeting other pathways of immune activation and inhibition.
Armored CAR T Cells
Adoptive T cell therapy is another emerging immunotherapeutic approach in ovarian cancer. Building on the MSK experience using chimeric antigen receptor (CAR) technology in hematologic malignancies, we are now investigating CAR T cell therapy in solid tumors. In our phase I ovarian cancer study NCT02498912, a patient’s own T cells are genetically modified to target the MUC16ecto tumor antigen and to secrete cytokine interleukin-12 (IL-12). (4)MUC16 is expressed in 70 to 80 percent of high-grade serous ovarian cancers and minimally expressed in normal tissues outside of the gynecological tract. The portion of MUC16 glycoprotein that is released into the blood stream is known as CA-125. Our CAR is specific for MUC16ecto, the retained portion of the glycoprotein that remains on the ovarian cancer cell surface. To overcome some of the inhibitory effects of the solid tumor microenvironment, our CAR T cells will be “armored” with the ability to secrete IL-12. This approach permits enhanced proliferation of the infused CAR T cells and augments the immune response at the tumor site. See Figure 2.

Patients eligible for CAR T cell immunotherapy have no history of autoimmune disease, have received two to five prior rounds of chemotherapy, and have tumors confirmed to express MUC16ecto. The first half of the CAR T cell dose is administered intravenously and the remaining cells are delivered through an intraperitoneal catheter into the abdominal cavity. Since there is a significant risk of toxicity related to cytokine release syndrome and IL-12, the first patients treated in the study will receive the CAR T cells in the intensive care unit, and all patients will be monitored closely. If successful, this technology could potentially be used as a therapeutic strategy for additional solid tumors by substituting the target tumor antigen. For example, CAR T cell technology targeting the tumor antigen mesothelin is under investigation in mesothelioma and breast cancer (NCT02414269).
