President Joseph Biden is one of an estimated 120,000 men living with stage 4 prostate cancer. Fortunately, there are effective treatments that can slow or stop prostate cancer from progressing, even when it has metastasized, or spread to other parts of the body, typically the bones.
“Patients with metastatic prostate cancer typically receive hormonal therapy as a first treatment,” says Memorial Sloan Kettering Cancer Center medical oncologist and Prostate Cancer Section Head Michael Morris, MD. “Even when the disease develops resistance to hormonal therapy, there are exciting new treatments that are effective.”
One new treatment uses radiopharmaceuticals to zero in on cancer cells to destroy them. The therapy, called 177Lu-PSMA-617 (Pluvicto®), includes a molecule that selectively seeks out and attaches to a specific protein on the cancer cell surface called PSMA (prostate-specific membrane antigen). It delivers radiation that damages DNA and destroys the cancer cell.
“At MSK, all patients with disease at risk for metastatic prostate cancer or who have relapsed disease on treatment are assessed by an imaging test, known as a PSMA-PET scan, that detects PSMA on the cells,” Dr. Morris says. “The patients who are resistant to hormonal therapy can receive 177Lu-PSMA-617 as part of their standard of care, and they are likely to feel better, function better, have less pain, and in some cases live longer than if they received an alternative.”
Recently, more patients became eligible for 177Lu-PSMA-617. In March 2025, the U.S. Food and Drug Administration expanded approval so it can be used earlier in treatment. Previously, the FDA had approved 177Lu-PSMA-617 only for patients with metastatic castration-resistant prostate cancer that had spread despite treatment with both an androgen receptor pathway inhibitor (ARPI) and taxane-based chemotherapy.
But results from a phase 3 trial led the FDA to expand the use of 177Lu-PSMA-617 to patients who have not yet received taxane therapy. It showed that 177Lu-PSMA-617 prolonged progression-free survival (the period when the disease did not progress) in people who received it compared with those given a second hormonal therapy The trial, called PSMAfore, was reported in The Lancet.
“The expanded FDA approval substantially increases the number of patients now eligible for treatment with 177Lu-PSMA-617,” Dr. Morris says. “This type of precision medicine is a game changer for people whose prostate cancer has spread outside of the prostate despite treatment with hormonal therapy,”
Dr. Morris was the Principal Investigator of the study in the US, and the lead author of the Lancet publication.
“A standard approach for patients with metastatic prostate cancer has been to give drugs that target the androgen receptor, and then if that fails, to give chemotherapy,” Dr. Morris explains. “But some doctors and patients have been delaying taxane therapy due to side effects, and instead switch to a different hormonal therapy. This trial suggests we can proceed straight to 177Lu-PSMA-617.”
Prostate cancer is the second leading cause of cancer death in American males and kills more than 35,000 people in the U.S. every year. The new therapy, developed by the pharmaceutical company Novartis, has been a breakthrough for treating prostate cancer after it has spread and grown resistant to other drugs.
Theranostics: An Emerging Field
The FDA approval is the latest bold advance in the emerging field of theranostics, which uses radioactive substances to visualize cancer cells and destroy them without harming normal cells. It also enables doctors to determine how well a treatment is actually working.
“We have a theranostic motto, which is ‘We see what we treat, and we treat what we see,’ ” says nuclear medicine physician Lisa Bodei, Director of Targeted Radionuclide Therapy at MSK. She is an expert specializing in using radioactive materials to diagnose and treat cancer and played a key role in the treatment of MSK participants in the trial.
The original clinical trial, called VISION, showed that adding the drug to standard treatment slowed progression of prostate cancer. The treatment was recognized as a breakthrough when Dr. Morris presented the results in June 2021 at the annual meeting of the American Society of Clinical Oncology. Those results also were reported in The New England Journal of Medicine.
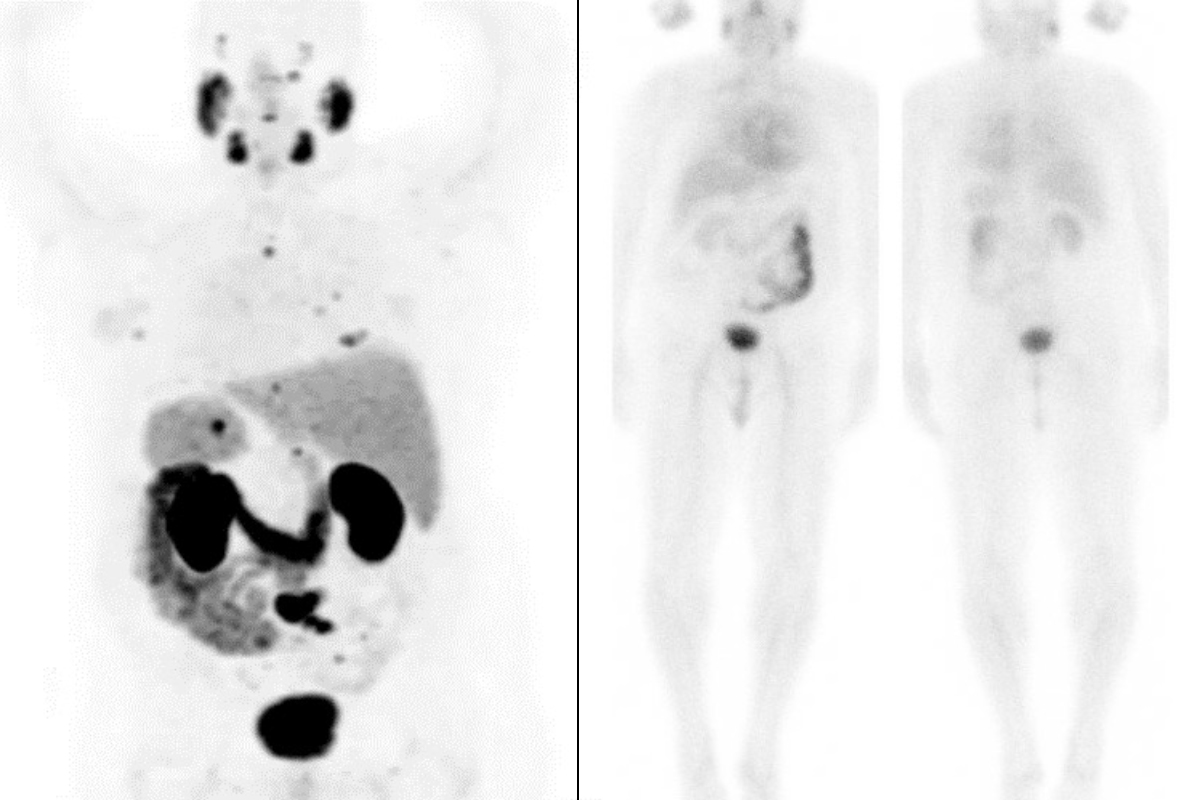
PSMA-PET scans of Michael Rosenblum before treatment (left) show prostate cancer metastases (small dark spots) throughout his body. After treatment (right), metastatic cancer is no longer visible.
A New Lifeline
The new therapy could be a lifeline for many people with metastatic prostate cancer. Just ask Michael Rosenblum. In 2019, his prostate cancer was resistant to chemotherapy and other treatments and had spread. PET scans showed dark clusters of cancer cells in bones throughout his body. His PSA levels — a marker that normally should be in the single-digit range — had soared to more than 100.
Dr. Morris enrolled Michael in the VISION clinical trial. Michael began treatment in July 2019 and ended in February 2020. After six doses of the therapy, follow-up scans showed the metastatic cancer was no longer visible. Five years later, the 79-year-old continues to be disease-free, with a PSA that is undetectable.
“I had no side effects either on the day of the procedures or afterward,” Michael says. “My PSA went right down, and my blood tests have been really good. From how I feel today, you would never think I had cancer a few years ago.”
How PSMA Lights Up Cancer Cells
In 2021, the U.S. Food and Drug Administration issued national approval to two new prostate cancer imaging tests based on similar technology. On a PET scan, the test lights up the cancerous cells that would otherwise be hidden, enabling doctors to precisely target treatment.
Both advances in imaging and therapy rely on targeting PSMA, which is not found on most normal cells but is overexpressed in cancer cells, especially those that have spread. The PSMA molecule was cloned at MSK in the early 1990s.
The Molecular Imaging and Therapy Service, led by Heiko Schöder, played a key role in the development and testing of a slightly different PSMA-directed imaging technology at MSK.
“This advance is the result of years of work by the community of physicians promoting the use of PSMA agents,” Dr. Schöder says. “It’s gratifying to see a collaborative effort result in a breakthrough that has the potential to make a difference for so many patients with advanced prostate cancer.”
Before receiving the therapy, patients are scanned with PSMA-directed PET imaging to make sure enough PSMA is present in the cells to make them likely to respond to the treatment. If so, they received the radioactive drug by injection over four to six sessions, spaced six weeks apart.
“I have been involved in the PSMA research since the end of my fellowship at MSK in the late 1990s,” says Dr. Morris, whose research has been supported by the philanthropy of John and Susan Magnier and Peter and Jean Scannell. “It’s amazing to see it all come to fruition. The benefits these advances will bring to men with this common disease cannot be overstated.”
Advances in radiotheranostics are supported by The Tow Foundation, long-time contributors to MSK’s mission.