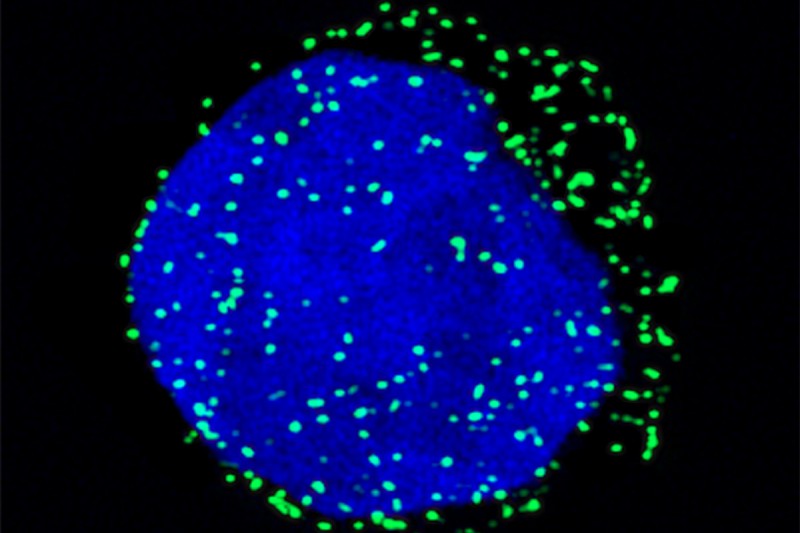
For patients with the blood cancers acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), the biggest barrier to a cure is relapse. Like a monster from a movie, the cancer rises up after being seemingly vanquished, often with fatal results.
Researchers think the problem lies with cancer stem cells that initiate and sustain the disease. These stem cells are highly resistant to standard therapies and — even after a seemingly successful treatment — go on to produce another crop of cancerous blood cells. It has become generally accepted that AML and MDS can be cured only if these stem cells are eradicated.
A new study by researchers at Memorial Sloan Kettering and NYU Langone Medical Center shows that a protein called CD99 appears in high levels on the surface of these cancer stem cells. This discovery, reported in the journal Science Translational Medicine, could prove to be an important step toward developing more-effective treatments for AML and MDS.
We spoke with MSK medical oncologist Stephen Chung, who led the study and collaborated with Christopher Park (who recently joined NYU Langone from MSK) and other researchers at MSK, Weill Cornell School of Medicine, and the University of Pennsylvania.
Why is the identification of CD99 as a prominent feature of these stem cells so significant?
While other surface proteins have been linked to these blood cancers, CD99 is distinctive in that it’s especially high in AML and MDS stem cells compared both with normal stem cells and with mature [fully formed] AML and MDS cells. This means we might be able to design therapies that selectively destroy this group of cells by targeting CD99. Also, being able to better isolate AML and MDS stem cells using CD99 will allow for improved studies of the origins of these diseases and their resistance to therapy.
In addition, we have shown that we can focus on CD99 to separate AML stem cells from normal stem cells in bone marrow taken from patients, and the technology already exists to potentially do this in the clinic.
How did you show that these CD99-rich stem cells are actually causing or sustaining AML or MDS?
In bone marrow samples taken from AML and MDS patients, we separated out the stem cells that had high levels of CD99 and put them into mice. The animals developed very aggressive and lethal leukemia. When we did the same thing with low-CD99 stem cells, they did not cause the disease, even when we injected them in very high numbers. That’s probably the most important finding: We were able to establish solid evidence that CD99 is a critical factor that can identify stem cells that sustain these diseases.
How could these findings help patients with AML and MDS?
In the shorter term, it could have important clinical applications for autologous bone marrow transplant. This is a treatment in which a patient’s own blood-forming stem cells are collected, the patient is treated with high-dose chemotherapy or radiation to kill the cancer cells, and then the collected stem cells are transplanted back into the patient, allowing the bone marrow to produce new blood cells. This approach has been tried in the past to treat some AML and MDS patients, but it hasn’t worked that well — we suspect it is because with this approach, we may be essentially giving patients back their own treatment-resistant stem cells, which initiate the cancer again. If we could use CD99 as a marker to purge the cancer stem cells from the bone marrow before it is donated, we could greatly reduce the risk of relapse.
Further down the road, this raises hopes that we can develop more effective therapies for these patients. We identified and generated antibodies that recognize and destroy CD99 stem cells while sparing normal stem cells, which we confirmed in experiments using patient cells growing in mice. The next step is to make a humanized form of these antibodies so this therapy can be taken forward into testing in the clinic.
How do the antibodies kill the high-CD99 stem cells?
When the antibody binds to CD99 on the stem cell surface, it appears to send a signal inside the cell that activates enzymes called SRC-family kinases. We think in normal cells, CD99 acts as a brake on this kinase activity, but when disabled by an antibody, the CD99 brake is removed and the kinases become overactive and cause the cell to self-destruct. This represents a therapeutic vulnerability in AML and MDS that may allow for the design of other therapies apart from the antibodies we have already identified. We hope all of these insights will lead to treatments that bring lasting remission or even a cure for people with these diseases.