
Metastasis researchers are keeping a close eye on the range of cell types that exist in a tumor’s local milieu, called the microenvironment, and on the molecules that cancerous and noncancerous cells secrete to communicate with one another.
“The microenvironment has a critical impact on a tumor’s ability to grow and metastasize,” said Johanna Joyce, who heads a laboratory in the Sloan Kettering Institute’s Cancer Biology and Genetics Program.
On the one hand, the influence of the microenvironment can suppress the aggressive behavior of tumor cells. For example, when breast tumor cells leave their primary location and infiltrate the lung, they usually perish from the encounter with a foreign environment, “which can be hostile to them since conditions are very different from those in the breast, where the tumor cells first developed,” Dr. Joyce explained.
But, she added, there are also scenarios in which tumor cells adjust and exploit their surroundings: “Some tumor cells can recruit noncancerous cells to a primary tumor or metastatic lesion, modify these cells, and co-opt them to stimulate their own growth or malignancy.”
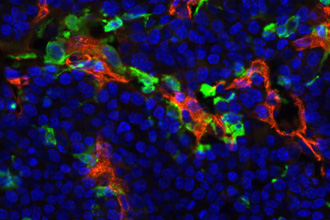
In a recent study published in Genes & Development, Dr. Joyce and her colleagues report on a mechanism they uncovered that allows tumor cells to communicate with white blood cells called macrophages, which are recruited to tumors from the bone marrow. [PubMed Abstract] By secreting a protein called IL-4, tumor cells can induce macrophages to produce protein-degrading enzymes called cathepsins. The enzymes’ activity helps tumor cells invade the surrounding normal tissue, which can set the stage for metastatic spread.
In mouse models of pancreatic cancer, the investigators were able to prevent invasive processes by blocking the activity of cathepsins produced by the macrophages. “Our findings show that there are opportunities for developing therapeutics directed against tumor-associated macrophages that could be very potent in controlling metastasis,” Dr. Joyce noted.
She and her colleagues are also exploring how such therapies could be used alongside drugs that kill tumor cells. Their research indicates that in response to some types of chemotherapy, cathepsin activity increases, “presumably increasing the risk that tumors will metastasize,” Dr. Joyce said.
“According to our preclinical studies, cathepsin inhibitors could be used to enhance the efficacy of our current chemotherapy treatments,” she added. “There is great promise in the development of combined therapies that target different components of the tumor microenvironment — for example, those that target tumor cells, white blood cells, and tumor-infiltrating blood vessels simultaneously.”