Lead Contacts
Grant Chen, MD
Takeshi Irie, MD, PhD
Department of Anesthesiology and Critical Care Medicine
Biomedical Engineering
Memorial Sloan Kettering Cancer Center
For more information contact
Anesthesiologists and biomedical engineers at MSK have modified the protective boxes now commonly employed during intubation of infectious patients, creating a two-piece setup instead of the usual single unit. This simple innovation allows medical professionals better access if difficulties arise during these procedures.
The one-piece boxes currently in circulation were devised to contain most of the aerosolized virus from spreading into the room, mitigating infectious exposure to the person intubating.
Our redesign adds a significant safety feature whereby the upper section can be removed (by an assistant) without the primary operator needing to remove their hands or the airway equipment from position.
Clinical Need
The novel coronavirus SARS-CoV-2 causes hypoxic respiratory failure resulting in the clinical entity COVID-19. There has been a massive increase in the need for ventilatory support of the most critically ill patients. To protect the anesthesiologist, ICU physician, ER provider, CRNA, or other healthcare providers against the aerosolized virus emitted when carrying out the intubation of such patients, hospitals across the country are increasingly using intubation boxes.
We believe our simple modification of going to a two-piece design will be very helpful in patients with difficult airways. Under normal circumstances around 2% of intubations are anticipated to be unexpectedly difficult. However, there have been reports (1) that patients with COVID-19 are presenting with edematous pharyngeal tissues (swollen airways), which may dramatically increase difficult intubations.
Innovation
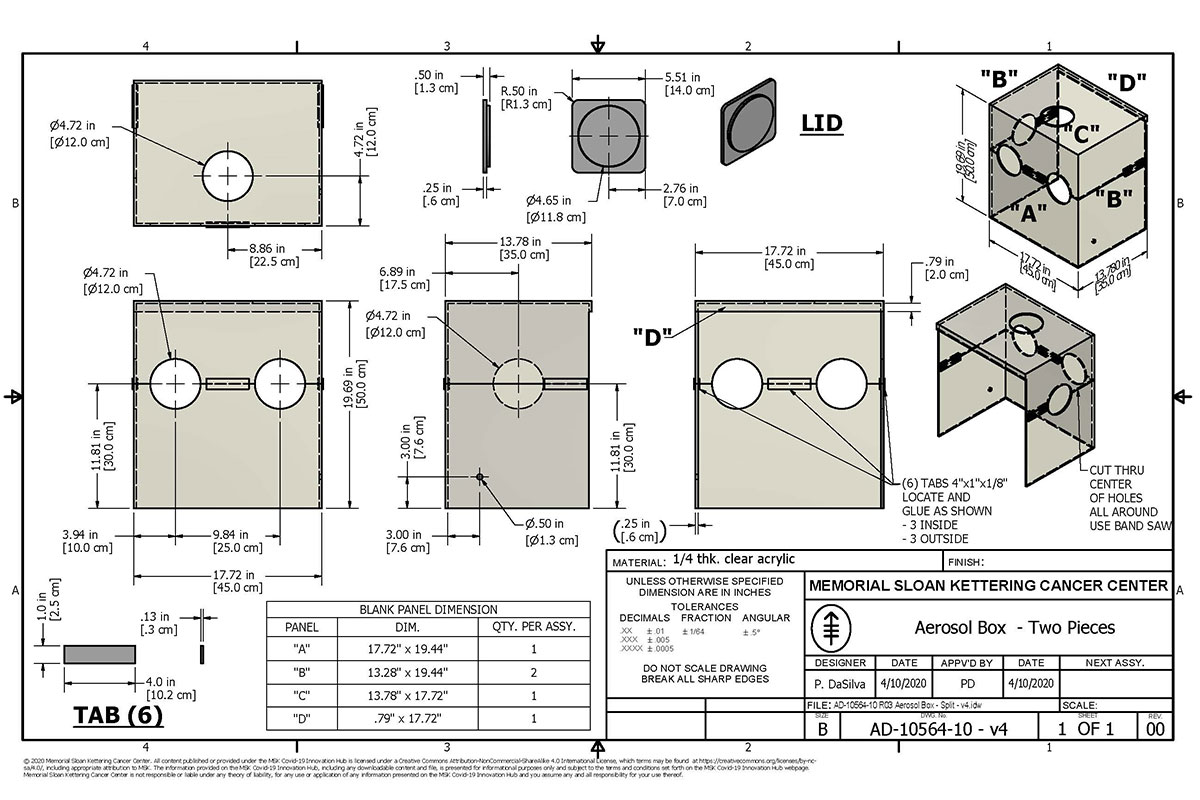
Along with the two-piece design we have incorporated holes on multiple sides of the box to allow access to the patient for all airway procedures such as intubation, bronchoscopy, and tracheostomy.
For further protection, and to reduce the number of aerosolized infection particles, a yankauer suction tube is placed inside the box and a sheet is draped on the caudal aspect of the box, creating a sealed chamber.
How to Make the Box
The box can be assembled from cut acrylic, plexiglass, or any other readily available transparent rigid plastic, glued to make the top and bottom pieces of the device. Semilunar “scoopouts” from the top and bottom form armholes for the operator. A rubber stopper covers the side hole. A small orifice on the right side allows for insertion of the suction tubing. The pieces are assembled around the patient at the time of use.
How to Use the Box
The box is placed over the patient’s head prior to intubation (or extubation), and intubation equipment is placed in the box, along with the anesthesia circuit. We recommend having the bag valve mask strapped to the patient to ensure full oxygenation prior to rapid sequence induction.
The intubator inserts arms into the holes to intubate. The plugged holes on the left and top allow easy access for a second person to provide cricoid pressure, insert additional instruments necessary for intubation, or to facilitate stylet removal.
In the event of difficult access to the airway, the assistant can immediately lift the lid while keeping the intubator’s hands in place.
While the position of the armholes is intended to be used for video laryngoscopy, we have successfully intubated patients using direct laryngoscopy with the armholes in these sizes and locations.
Cleaning
- Using the purple Sani-cloth wipes, wipe down the surface inside the procedure room, with complete removal of mucus, blood, and visible secretions
- Wipe the surface again, outside the procedure room
- Allow at least two minutes prior to reuse (we prefer 10).
References
- https://www.youtube.com/watch?v=GLbKyc31XhM skip to 21:48 to see anecdotal report of frequent pharyngeal edema by Dr. Anand Swaminathan of St Joseph’s Hospital in Paterson NJ
- https://sites.google.com/view/aerosolbox/design?authuser=0
Contributors
Grant Chen, MD, Anesthesiology and Critical Care Medicine
Takeshi Irie, MD, PhD, Anesthesiology and Critical Care Medicine
Sam Hellman, PhD, Biomedical Engineering/Medical Physics
Alexander J. Tanchoco, Digital Informatics and Technology Solutions
Emily Winograd, Strategy and Innovation
Alice Ro, Strategy and Innovation
Paul Booth, MS, Medical Physics
Gregory W. Fischer, MD, FASA, Chairman, Anesthesiology and Critical Care Medicine
The MSK COVID Safety Innovations Team
