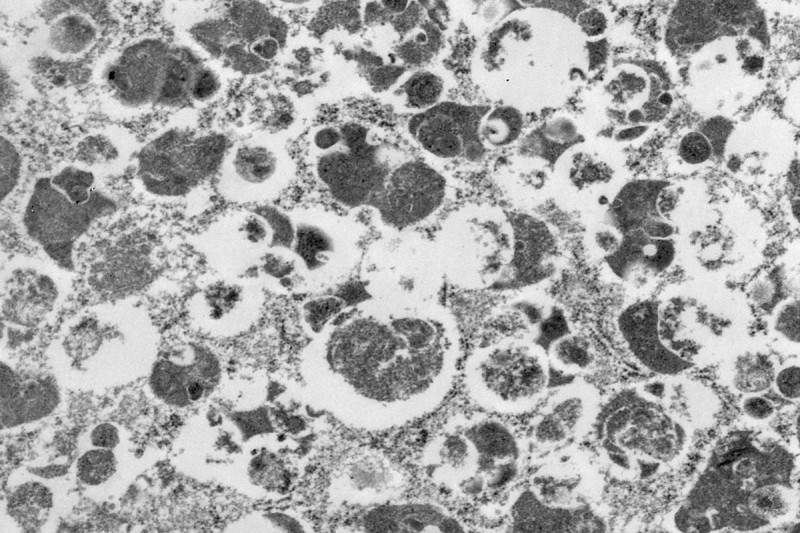
Granular cell tumors are rare growths that can occur anywhere in the body. The vast majority are not cancerous, but for the small number of people who have cancerous granular cell tumors, the rate of survival is poor. An enduring mystery about these tumors concerns the tiny granules within cells that give them their name. Researchers have not known what causes the granules to form, what function they may have, and if they play a role in causing the tumor growth.
Memorial Sloan Kettering researchers took a close look at the genetic makeup of granular cell tumors. They hoped to uncover the so-called driver mutations that trigger the abnormal growth. Led by experimental pathologists Jorge Reis-Filho and Britta Weigelt, the team sequenced every gene in 17 tumors and made a surprising discovery. Although the tumors did not have genetic changes in known cancer-related genes, nearly three-fourths of them had mutations in either the ATP6AP1 or ATP6AP2 genes.
The team validated the finding by sequencing an additional 64 granular cell tumors. Again, three-fourths of the tumors had mutations in these genes. The mutations were mutually exclusive — tumors had changes in either ATP6AP1 or ATP6AP2, but not both — and always resulted in the loss of the gene’s function.
“Neither of the genes has been linked to cancer in any way,” Dr. Reis-Filho says. He points out that very large-scale studies — such as The Cancer Genome Atlas, a multicenter initiative that has analyzed the biggest set of tumor samples to date — have not implicated these genes in cancer. For common cancers, mutations that cause the loss of ATP6AP1 or ATP6AP2 were found in less than 0.1% of tumors.
“The high frequency of these mutations in such a high percentage of granular cell tumors and their mutually exclusive nature are strong lines of evidence that they are driving the tumor growth,” Dr. Reis-Filho says. “It also suggests that we have discovered two genes that can act as tumor suppressors.”
The finding is reported today in Nature Communications.
Linking Control of pH Levels and the Development of Cancer
ATP6AP1 and ATP6AP2 have two critical functions. One is controlling pH levels in cells. These levels need to be closely regulated so proteins can function properly. The ATP6AP1 and ATP6AP2 genes also control vesicle trafficking. This is a biological process through which small structures transport materials between different compartments within a cell.
To test what would happen when these genes lost their normal function, the researchers disabled them in normal human cells in a lab dish. If either gene was silenced, the cells developed the same granules found in human granular cell tumors.
“Because ATP6AP1 and ATP6AP2 control pH and trafficking inside cells, removing the function of either gene causes the vesicles to pile up and form granules,” Dr. Weigelt explains.
The research team also showed that the loss of function of either gene could cause normal cells to begin forming tumors. The process through which this happens is not yet clear.
In the short term, knowing the link between ATP6AP1 and ATP6AP2 mutations and granular cell tumors could help with diagnosis. A simple sequencing test for these mutations in the tumor cells would do the trick.
In the longer term, clarifying what drives tumor growth could help with the development of therapies.
“This study is the first to link genetic alterations in genes that control pH inside a cell and the development of cancer,” Dr. Weigelt says. “These results provide new avenues for the use of targeted agents for the treatment of people with aggressive forms of granular cell tumors.”
Insights from Rare Cancers
The finding highlights the usefulness of studying rare cancers to identify the role that new genes play in the disease. Common cancers tend to involve a wide array of mutations. That means a large number of tumors need to be sequenced to identify new driver mutations. In contrast, rare cancers are often caused by a small number of driver mutations. These can be discovered more quickly by sequencing a handful of tumors. Dr. Reis-Filho’s laboratory demonstrated this in 2014 by identifying a new driver mutation in a rare form of salivary gland cancer called polymorphous low-grade adenocarcinoma.
“When we find a driver mutation in a rare cancer, we can test whether it applies in the more common cancers — at least specific subtypes of those cancers,” Dr. Reis-Filho says. “If we find a mutation, we can quickly develop genetic diagnostic tests for them as well.”
This study was funded by MSK’s Functional Genomics Initiative and the Breast Cancer Research Foundation.
