The Bakhoum lab focuses on understanding the role of chromosomal instability (CIN) in tumor progression and evolution. CIN is a hallmark of human cancer, and it is associated with poor prognosis, metastasis, and therapeutic resistance. It results from errors in chromosome segregation during mitosis, leading to structural and numerical chromosomal abnormalities. In addition to generating genomic heterogeneity that acts as a substrate for natural selection, our team has found that CIN promotes pro-tumor inflammatory signaling by introducing genomic double-stranded DNA into the cytosol, engaging cytosolic DNA-sensing antiviral innate immune pathways. In addition, we discovered that chromosome encapsulation in micronuclei can be subject to significant changes in histone post-translational modifications leading to abnormalities in chromatin accessibility. These multipronged effects distinguish CIN as a central driver of tumor evolution and as a genomic source for the crosstalk between the tumor and its microenvironment.
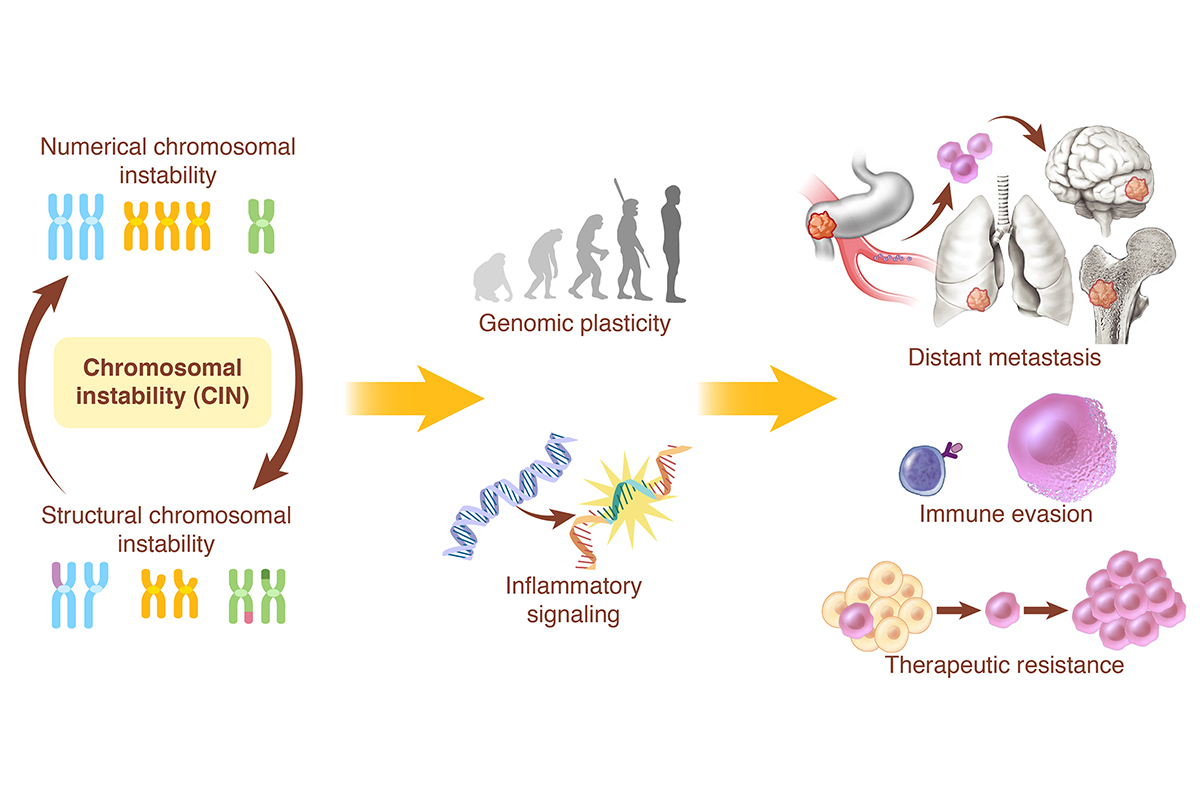
To better understand how CIN influences tumor evolution, we aim to deconstruct the individual cellular and microenvironmental phenotypes imparted by chromosome segregation errors. These phenotypes include genomic copy number heterogeneity, epigenetic abnormalities as well as inflammatory signaling, all of which can have distinct phenotypes on the tumor and its microenvironment. By understanding these basic processes, we aim to develop CIN-directed therapeutic strategies for the treatment of advanced cancers.