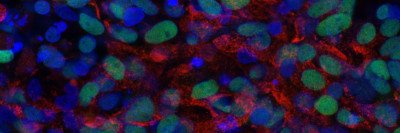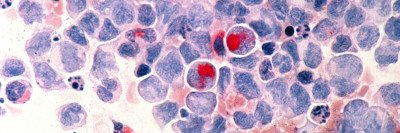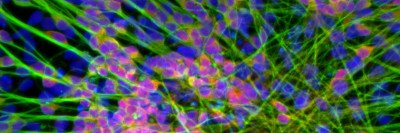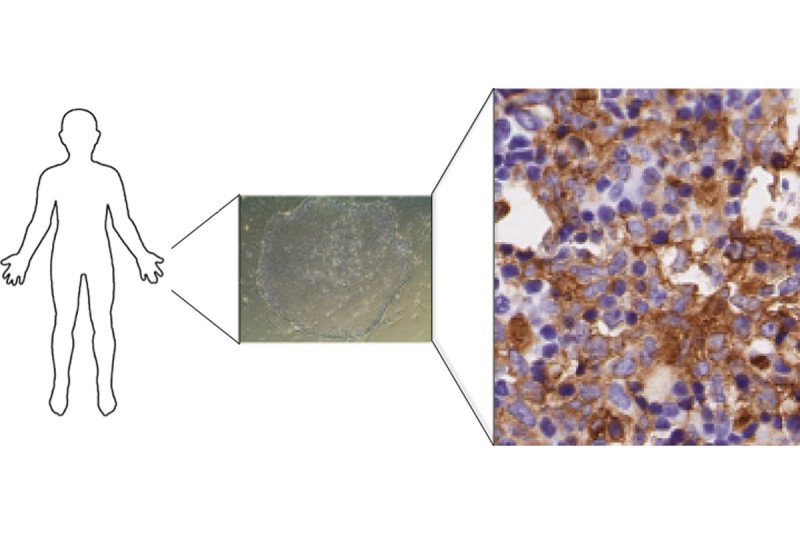
Like many cancers, acute myeloid leukemia (AML) moves through a step-by-step progression as it develops. It begins in normal blood cells, which then accumulate mutations that cause a group of blood diseases called myelodysplastic syndromes (MDS). Further mutations drive the cells to become full-blown AML.
In a new study, researchers from Memorial Sloan Kettering — in collaboration with a team from the Mount Sinai Icahn School of Medicine in New York City — report that for the first time, they’ve been able to produce a series of models that replicate each stage of the disease. This provides an important new tool for developing drugs to treat AML, as well as for finding ways to control and manage other pre-leukemic blood diseases like MDS.
“What we’re describing in this study is a new platform for studying leukemia and how it develops,” says MSK cancer biologist Michael Kharas, one of the two senior authors of the new research, published February 16 in the journal Cell Stem Cell. “We now have a way to look at leukemia that is stage-specific and to study all the potential genetic mutations that contribute to the disease.” The other senior author and leader of this study was Eirini Papapetrou, a cancer and stem cell researcher at the Tisch Cancer Institute at Mount Sinai.
Making Stem Cells from Cancer Cells
The scientists took an innovative approach to this research, using cancerous and precancerous cells from patients to generate what are called induced pluripotent stem cells (iPSCs). Such iPSCs — which have their clocks turned back to the earliest stages of development — are becoming an increasingly important research tool because they have the potential to develop into any of the specialized cell types found in the body.
Normally, researchers produce these cells from healthy cells, not cancerous ones. “The ability to make iPSCs from adult cells has been around for about a decade, but making them from cancer cells is still challenging,” Dr. Kharas explains. “What’s impressive is that the Papapetrou laboratory was able to make these various cells from [people with MDS and AML], as the genes in these cells are very screwed up, which makes it more difficult for the process to work.”
Once the researchers at the Papapetrou laboratory created the iPSCs from the cancer cells, they exposed them to a variety of normal developmental signals that caused them to begin developing into a number of different types of cells found in the body. Although the IPSCs were derived from cancer cells, they appeared normal before they were induced to develop into other cell types. However, when the investigators coaxed them into becoming blood cells, they took up the characteristics of cancer cells again.
“The cells that had been taken from people with AML became AML cells; the cells that had been taken from people with low-risk MDS became low-risk MDS cells; and so on,” Dr. Kharas says. “Han Yuan, a graduate student in the laboratory of MSK computational biologist Christina Leslie, was able to demonstrate that the gene expression programs resembled the patients with the disease.”
“This gave us a spectrum of different diseases that we can model and do further experiments with,” he adds. “Our contribution was that we were able to transplant the AML cells into mice and get a leukemia model that accurately represents what is seen in patients — something that has never been done before.”
Creative Gene Editing
Once the researchers created their library of cells, they used a gene-editing approach called CRISPR/Cas9 to do further experiments.
“Using this powerful technique, we can introduce new gene alterations or correct existing ones and move the cells back and forth along the different stages of leukemia development using the ‘phenotypic map’ that we constructed,” Dr. Papapetrou says. “We can also use these cells to test if specific drugs will elicit a better response in cells with specific genetic changes.”
Although the research is still in its early stages and has not yet led to any specific drugs to be tested in patients, the researchers are continuing to work with these models. In addition, they believe the techniques used in this study may also be applicable to studying how other cancers, including solid tumors, progress through various stages of disease.
This research was funded by the National Institutes of Health under grants R01HL121570 and R00DK087923, the Damon Runyon Cancer Research Foundation, the Edward P. Evans Foundation, the Ellison Medical Foundation, the Henry and Marilyn Taub Foundation, the Babich Family Foundation, and Alex’s Lemonade Stand Foundation.