Tumors of the pea-size pituitary gland, which sits at the base of the brain and secretes hormones, are very common. As many as one in five Americans may have a pituitary tumor without knowing it because the tumors don’t cause symptoms. In some people, benign (noncancerous) tumors can create serious problems by making excess hormones that can cause diabetes, mood changes, sleeping problems, gigantism, and other conditions. But even these tumors are usually treatable with surgery or medication.
A small number of pituitary tumors are aggressive and even cancerous, however. They grow quickly, typically come back after surgery and radiation treatment, and can even spread throughout the body (metastasize). In the past, people with these aggressive tumors have not had effective treatment options, or much cause for hope. The median survival of people whose pituitary tumors have metastasized is only one year.
Despite this grim outlook, doctors suspected that people with pituitary tumors might benefit from immunotherapy drugs called checkpoint inhibitors. The drugs have been effective against a variety of cancers, including melanoma, lung cancer, sarcoma, and lymphoma. People who are treated with checkpoint inhibitors for these cancers often have pituitary inflammation as a side effect.
“This type of inflammation is otherwise very, very rare,” says MSK neuroendocrinologist Eliza Geer, Medical Director of MSK’s Multidisciplinary Pituitary and Skull Base Tumor Center. “It suggests that checkpoint inhibitors may be targeting proteins in the pituitary cells. This brought up the possibility that pituitary tumors might respond to these drugs.”
A First in Immunotherapy Treatment
In December 2017, Dr. Geer and MSK neuro-oncologist Andrew Lin decided to use immunotherapy to treat a woman with an aggressive pituitary cancer. The patient, in her 40s, was out of options. She had already been through multiple surgeries, radiation treatment, and chemotherapy over the previous few years. Nothing had worked for long.
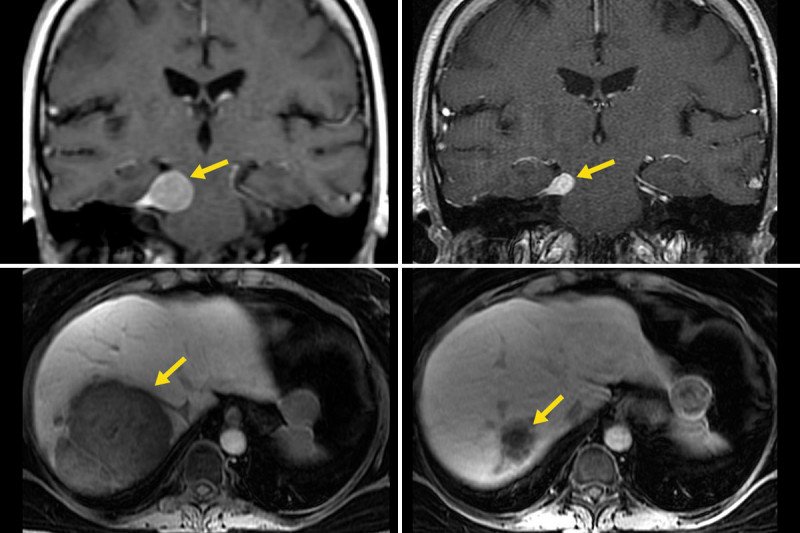
The doctors gave the woman a combination of two checkpoint inhibitors: ipilimumab (Yervoy®) and nivolumab (Opdivo®). The results were striking. Both the primary tumor in the pituitary gland and a large metastatic tumor in the liver shrank markedly.
“It’s really exciting to see such a response in this kind of aggressive tumor,” says Dr. Lin. “Typically, the most you can hope for is to stop the growth. To see such a dramatic response with so much shrinkage is unheard of.”
This woman’s treatment marks the first reported use of immunotherapy against pituitary tumors. It was reported this month in the Journal of Clinical Endocrinology and Metabolism. Based on this result, a clinical trial testing the approach in more people is expected to launch before the end of the year.
Even before the tumors shrank, there were other clear signs that the drugs were working for the patient. Her pituitary tumor was a type that makes an excess of a hormone called ACTH. After just one week, her ACTH level had decreased tenfold.
“The drop in ACTH provided more evidence that the drugs were affecting the tumor’s activity, giving us added incentive to investigate this further in a clinical trial,” Dr. Geer says.
The patient is continuing to take nivolumab alone, which is the standard immunotherapy approach used for other cancers treated with checkpoint inhibitors.
Mutational Patterns
In addition to the encouraging treatment result, Drs. Geer and Lin also learned important information about genetic changes that may develop in response to chemotherapy. A sample of the primary tumor was taken before the woman received chemotherapy, and a second sample was taken from the tumor that had spread to her liver. Both were analyzed using MSK-IMPACT™. This powerful sequencing technology looks for mutations in 468 cancer-related genes.
The liver tumor had a high number of mutations that the primary tumor didn’t have. This genetic pattern is called hypermutation. It has been detected in many cancers treated with chemotherapy. Researchers believe that hypermutation in the woman’s pituitary tumor was caused by chemotherapy.
“That’s another important aspect we want to investigate further — whether tumors with hypermutation are especially responsive to immunotherapy,” Dr. Lin says.
Testing in More People
The phase II clinical trial will test the ipilimumab and nivolumab combination in ten or 11 people and include genetic analysis of the tumors. If the responses are good, the trial may expand. What began with one person could lead to important insights that could help many people with pituitary tumors and other cancers.
“Our group here is uniquely positioned to do this kind of study because we have an integrated approach to the treatment of patients with pituitary tumors,” Dr. Geer says. “We have pituitary endocrine expertise, surgical expertise, neuro-oncology expertise, and the ability to do genomic analysis. The trial is in a preparatory phase, and we will make an announcement once it is open. We hope to get referrals from other hospitals in the area because no one else is doing this.”
The Multidisciplinary Pituitary and Skull Base Tumor Center is a unique, collaborative clinic. Patients are seen by neuroendocrine, neurosurgical, and head and neck experts. The team holds a regular pituitary and skull base tumor board, during which doctors work together to provide patients with the best care plan and advice on advanced therapies.