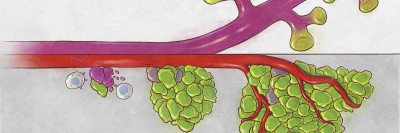
The standard treatment for men with advanced prostate cancer is androgen-deprivation therapy. Androgens are hormones that fuel prostate cell growth; removing them with either drugs or surgery causes the prostate gland to shrink by 90%.
Nevertheless, the cells that remain can eventually regrow a tumor, and when they do, the tumor is usually resistant to further hormone therapy. It is also more likely to spread to other organs (metastasize).
A new study from researchers at Memorial Sloan Kettering provides insight into how the prostate is able to regrow so swiftly. And it is not what the scientists initially expected.
“Most people, including me, expected to find a rare population of stem cells that is responsible for regenerating the gland,” says Charles Sawyers, Chair of the Human Oncology and Pathogenesis Program at MSK and the corresponding author on the paper, published May 1 in the journal Science. “But this is not the case.”
Instead, he says, nearly all of the cells that persisted after hormone-deprivation therapy contributed to the regeneration of the prostate gland. Most of these were luminal cells, which form the inside of the hollow organ. The findings have implications for how doctors think about prostate cancer treatment.
A Cellular Atlas of the Prostate
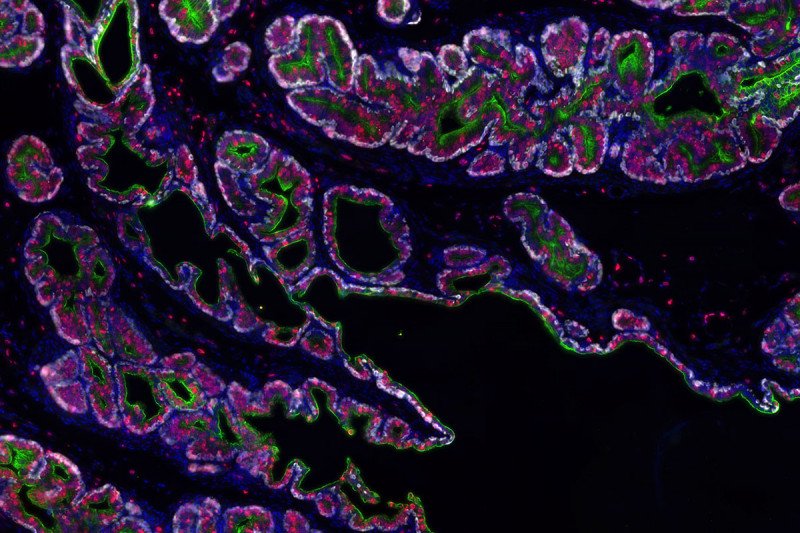
The investigators made their discovery with the help of a powerful technique called single-cell RNA sequencing (scRNA seq). This method of analysis allows scientists to identify which genes are turned on in many individual cells in a tissue at once. Collaborating with scientists from the Dana Pe’er lab in the Sloan Kettering Institute and Aviv Regev’s lab at the Broad Institute, the team performed scRNA seq on nearly 14,000 cells in the mouse prostate gland. From these data, they were able to completely map out the cell types found in a normal mouse prostate.
With the information, they could then determine which cell types remained in the prostate after the mice received androgen-deprivation therapy, and which divided to regrow the gland when androgen was restored.
From these analyses, it was clear that nearly all the luminal cells in the prostate were dividing, rather than just a subset — as would be expected if a stem cell population were mainly responsible for regenerating the gland.
What’s more, the luminal cells (whose typical function is to secrete fluids) had clearly acquired abilities they don’t usually have in a hormonally intact animal.
“They became much more stemlike,” says Wouter Karthaus, a senior postdoctoral fellow in the Sawyers lab and the paper’s first author. “Without androgens influencing their gene expression, they were free to turn on other genes and acquire regenerative properties.”
In addition to the mouse work, the investigators performed scRNA seq on prostate tissue taken from men who had been treated for prostate cancer. They found a similar pattern of luminal prostate cells that had acquired the attributes of stem cells. This implies that what is true of mice may also be true of men.
Plastic Identities
The study’s findings contradict a classic model of how stem cells regenerate and repair tissue. By that way of thinking, stem cells are a rare and special type of cell that can give rise to many cell types yet retain a proliferative capacity. But recent studies — including, now, this one — have questioned how broadly this model applies across different organs. At least in the prostate, fully differentiated cells can become stem cells under the right conditions, this study suggests.
Prior work from the Sawyers lab showed that some prostate cancer cells possess an ability to change their identity. This is called lineage plasticity. They can, for example, reprogram themselves to become a type of prostate cell that does not require androgen to survive. Lineage plasticity is an important way that prostate cancer cells eventually develop resistance to hormone-blocking therapies.
What the latest findings mean for treatment is an open question, but there could be significant implications.
“Androgen-deprivation therapy may be a double-edged sword,” Dr. Sawyers says. “Lots of cells die, but the ones that persist acquire this stemlike property.”
“It is likely that we push prostate cancer to have a more progenitor-like state during therapy,” Dr. Karthaus adds.
The team’s next effort will be to identify the molecular and cellular cues that control this switch in the hope of developing ways to turn it off.
The study was supported by the Howard Hughes Medical Institute, National Institutes of Health grants (CA193837, CA092629, CA224079, CA155169, and CA008748), a Starr Cancer Consortium grant (I12-0007), the Klarman Cell Observatory, National Cancer Institute (NCI) grants (1U24CA180922 and R33-CA202820), the Koch Institute NCI core grant (P30-CA14051), the Ludwig Center at the Massachusetts Institute of Technology, the Dutch Cancer Society, and a Prostate Cancer Foundation Young Investigator Award.
Dr. Sawyers is on the board of directors of Novartis, is a co-founder of ORIC Pharmaceuticals, and is a co-inventor of the prostate cancer drugs enzalutamide (Xtandi®) and apalutamide (Erleada®), covered by US patents 7,709,517; 8,183,274; 9,126,941; 8,445,507; 8,802,689; and 9,388,159, filed by the University of California. He is on the scientific advisory boards of the following biotechnology companies: Agios, BeiGene, Blueprint, the Column Group, Foghorn, Housey Pharma, Nextech, KSQ Therapeutics, Petra Pharma, and PMV Pharma. Dr. Sawyers is a co-founder of Seragon Pharmaceuticals, purchased by Genentech/Roche in 2014.
Dr. Karthaus is a co-inventor on patent WO2012168930A2, filed by the Royal Netherlands Academy of Arts and Sciences, which covers organoid technology. Dr. Regev is a co-founder of and equity holder in Celsius Therapeutics, is an equity holder in Immunitas, and is on the scientific advisory boards of Syros Pharmaceuticals, Neogene Therapeutics, Asimov Biotechnology, and Thermo Fisher Scientific.