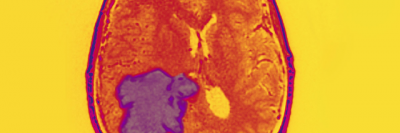
Neurofibromatosis type 1 (NF1) is the most common hereditary disease of the nervous system. Overall, it is a rare condition that affects about one in 3,000 people in the United States and worldwide. It is characterized by tumors that grow along the peripheral nerves (the nerves outside the brain and spinal cord) as well as changes in skin pigmentation.
For some people with NF1, their tumors may become cancerous, including a type of sarcoma called malignant peripheral nerve sheath tumor (MPNST), which usually arises in childhood. In a study published online May 18, 2021, in Cell Stem Cell, researchers from Memorial Sloan Kettering reported they have discovered that a subpopulation of cells within these tumors could explain why the tumors almost always come back after treatment. The research, which was carried out in mice, suggests that scientists eventually could be able to create targeted therapies for the treatment of people with MPNST.
Inside My Lab: Luis Parada
“Over the past 20 years or so, we’ve been developing animal models for NF1,” says developmental biologist Luis Parada, the senior author of the paper. “One reason is that it is a disease that is very hard to treat, and more research is needed. Another reason is that we know it’s caused by a single gene mutation, which makes it somewhat easier to study than diseases with more complex causes.”
Linking Early Development to Cancer
Dr. Parada, who is Director of MSK’s Brain Tumor Center and a member of the Cancer Biology and Genetics Program in the Sloan Kettering Institute, focuses his research on cancers of the nervous system. This includes different kinds of brain tumors as well as tumors like MPNST that affect the cells that protect the nerves outside the brain and spinal cord.
“A large part of the work done in my lab is studying how the nervous system develops,” he says. “The concept that has emerged in recent years is that cancers of the nervous system are connected to specific cell lineages related to early development.” In other words, these cancers arise from nerve stem cells, which are building block-like cells that can take up any form. Understanding how and why this happens is important to finding better therapies.
Dr. Parada’s lab has developed a number of mouse models to study nervous system tumors. Some, like the mice in this research, are genetically engineered to have mutations that cause cancer to grow. Others, called xenograft models, have tumors implanted in them from human patients.
Figuring Out Why Cancer Returns
About 25% of people with NF1 develop tumors called plexiform neurofibromas, which can become very large but are not classified as cancerous because they don’t spread to other parts of the body. However, between 5 and 10% of plexiform neurofibromas evolve into cancerous MPNSTs.
One thing that makes MPNSTs so challenging to manage is that even when treated — either by surgery, radiation, chemotherapy, or some combination of these therapies — they almost always come back and spread to other parts of the body. In the current study, the investigators did several experiments to look at why this is the case.
In one experiment, they gave the mice chemotherapy and looked at the cells that remained in the tumor after treatment. They also labeled specific tumor cells with a green fluorescent protein that allowed them to follow where they traveled in the body. Some cells were also labeled with a chemical that allowed the research team to determine how quickly the cells were dividing.
The scientists used these tools to study how the cancer in the mice grew back. They found that two types of cells were contributing to tumor formation: cells that divide rapidly, a behavior typical of many cancer cells, and cells that were quiescent, or quiet and slow growing. The quiescent cells appeared to be very similar to neural crest cells, a type of stem cell that gives rise to many kinds of nerve cells, which helped to reveal what was different about these cells.
“Chemotherapy and radiation target cells that are dividing rapidly,” Dr. Parada explains. “But quiescent cells are not affected by the treatment. This research explains why chemotherapy and radiation alone are not sufficient to completely eliminate the cancer.”
Seeking a Combination Approach
Ultimately, this research suggests why treatments that go after fast-growing cells are insufficient to destroy MPNSTs, Dr. Parada says. The quiescent cells that remain after chemotherapy have the capacity to regrow the tumor. Treatments that combine chemotherapy with targeted agents that destroy quiescent cells are much more likely to be successful.
One of the next steps for this research is to validate the findings that were made in genetically engineered mice using patient samples. Dr. Parada’s team will also use patient-derived xenograft models to continue studying the behavior of these tumors.
One of the co-authors on the current paper is Sameer Farouk Sait, a researcher in Dr. Parada’s lab who is also a pediatric oncologist at MSK and is focused on applying these findings to clinical practice. Dr. Parada’s lab is also participating in an ongoing collaboration with labs at several other institutions to study tumors caused by mutations in the NF1 gene. This effort is funded by a National Institutes of Health (NIH) Specialized Programs of Research Excellence (SPORE) grant.
In addition to the SPORE grant (U54 CA196519-01), this research was funded by NIH grants CA131313 and P30 CA008748. Dr. Parada is a recipient of an Investigator-Initiated Research Award in Congressionally Directed Medical Research Programs from the Department of Defense (grant W81XWH-16-1-0186) and holds the Albert C. Foster Chair in Cancer Research at MSK.