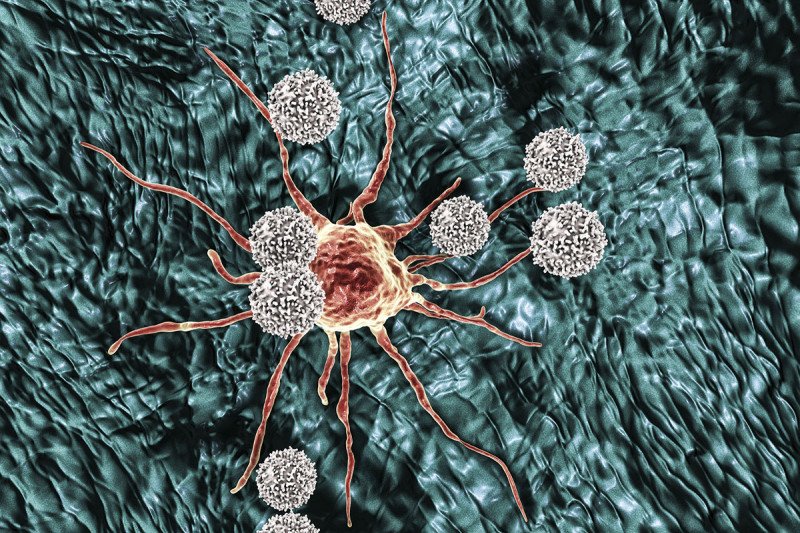
Perhaps the biggest challenge facing cancer immunotherapy is figuring out why it doesn’t work for everyone. Currently only 20% to 40% of people respond to these treatments, which harness the power of immune cells (T cells) to attack tumors. A major hurdle appears to be something called T cell exhaustion.
T cells seek and destroy cells that have molecular markers on their surface, which the immune system recognizes as a foreign signal. These markers are called antigens. But when T cells sense the same antigen signal for extended periods they become “exhausted” and lay down their arms. This prevents the immune system from attacking for too long and harming the body’s own tissue.

“The T cells turn on the brakes to tamp down their own activity,” says physician-scientist Santosha Vardhana, a researcher in the Craig Thompson laboratory at the Sloan Kettering Institute.
This phenomenon explains why T cells often flock to tumors and invade them, only to then lose their power. Cancer immunotherapies called checkpoint inhibitors work when they manage to release the brakes on T cells and prod them back into action. But for most people, the immune cells stay dormant. Understanding exactly how T cell exhaustion develops could be critical for making immunotherapy treatments work for more people.
A new study led by Drs. Vardhana and Thompson has found that T cell exhaustion starts with the cells’ metabolism — the chemical processes that enable cells to produce energy from nutrients. These changes in metabolism send T cells down the path to exhaustion.
“We’ve shown that if you really want to rewire a T cell so it can be better at killing tumors, you have to change its metabolism,” Dr. Vardhana says.
The finding is published in Nature Immunology.
The Source of the Problem
The idea that metabolism might play a critical role arose after studies showed that a successful response to immunotherapy depends on whether the T cells maintain their ability to divide and make new cells (proliferate). Nearly 20 years ago, Dr. Thompson and colleagues showed that T cells require a shift in their metabolic behavior to proliferate.
Dr. Vardhana and his collaborators therefore wondered whether loss of the ability to proliferate in T cells from patients who do not respond to immunotherapy might result from a defect in the cells’ metabolic fitness. But it was hard to tell if this was the case because researchers had largely been looking at T cells taken out of tumors. By then, the T cells were both exhausted and metabolically inactive, and it was hard to determine cause and effect.
“It was like reading the last chapter of a book and trying to figure out what the main conflict was,” Dr. Vardhana explains.
To understand this, the research team built a model system that allowed them to tease apart the process of T cell exhaustion. In a dish, they continuously exposed T cells to foreign signals from tumor antigens and watched the reaction over time. They found that continuous exposure to tumor antigens overtaxed a part of the T cell called the mitochondrion — otherwise known as the “powerhouse” of the cell — which is responsible for converting nutrients into energy.
Unable to shoulder the load imposed by persistent exposure to tumors, the mitochondria within T-cells begin to leak molecules called free radicals, which block essential cellular processes — including the ability to proliferate — and damage different parts of the cell, including the mitochondria themselves.
“Over time, T cells in tumors get trapped in a vicious cycle,” Dr. Vardhana says. “Damaged mitochondria leak even more free radicals, which further damage the mitochondria.” Of note, Dr. Vardhana and colleagues saw signs of free radical damage early on, suggesting that this metabolic abnormality is a cause, rather than a consequence, of T cell exhaustion.
The researchers subsequently tried to block the damage. They dosed T cells with antioxidants, which soak up free radicals. This seemed to work, allowing T cells to proliferate and kill tumor cells indefinitely.
“What was even more surprising was that in addition to recovering T cell function, antioxidants seemed to essentially move the T cell ‘backwards in time’ on the exhaustion trajectory — from a more exhausted cell to a less exhausted cell,” Dr. Vardhana says. “We are still trying to understand how a T cell senses a change in metabolism and reacts by changing its entire cell state.”
Ultimately, Dr. Vardhana and colleagues feel that this discovery could be an important step toward making cancer immunotherapies work better. “Knowing what lies at the heart of T cell exhaustion gives us a strategy for making all these immunotherapies work for more people,” Dr. Vardhana says.
Dr. Vardhana is a Senior Fellow with the Parker Institute of Cancer Immunotherapy and is supported by a Burroughs Wellcome Fund Career Award for Medical Scientists. This work was additionally supported by the Memorial Sloan Kettering Cancer Center Support Grant P30 CA008748 and R25 Training Grant AI140472-01A1.
Dr. Vardhana has received honoraria from Agios Pharmaceuticals and Rheos Pharmaceuticals, is an advisor for Immunai, and has consulted for ADC Therapeutics. Dr. Thompson is a founder of Agios Pharmaceuticals and a member of its scientific advisory board. He is also a former member of the Board of Directors and stockholder of Merck and Charles River Laboratories.