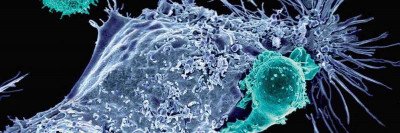
What is CAR T cell therapy?
Chimeric antigen receptor (CAR) T cell therapy is a type of therapy that uses your immune system (immunotherapy). Your immune system’s first job is to help your body fight infections. T cells (white blood cells that are part of the immune system) tell your body what it should fight. T cells usually fight infections, but they can also fight cancer. CAR T cell therapy uses your child’s own T cells to find and attack cancer cells.
How does CAR T cell therapy find cancer?
Chimeric antigen receptor (CAR) T cell therapy is a type of immunotherapy. We change T cells in a laboratory so they will attack cancer cells.
Antigens are on the surface of cancer cells. T cells have receptors that attach to certain antigens. Once a T cell attaches to an antigen, it can kill that cell. The CAR is a receptor that helps T cells find and kill cancer cells. We will change T cells in a lab by adding the CAR receptor. They become CAR T cells.
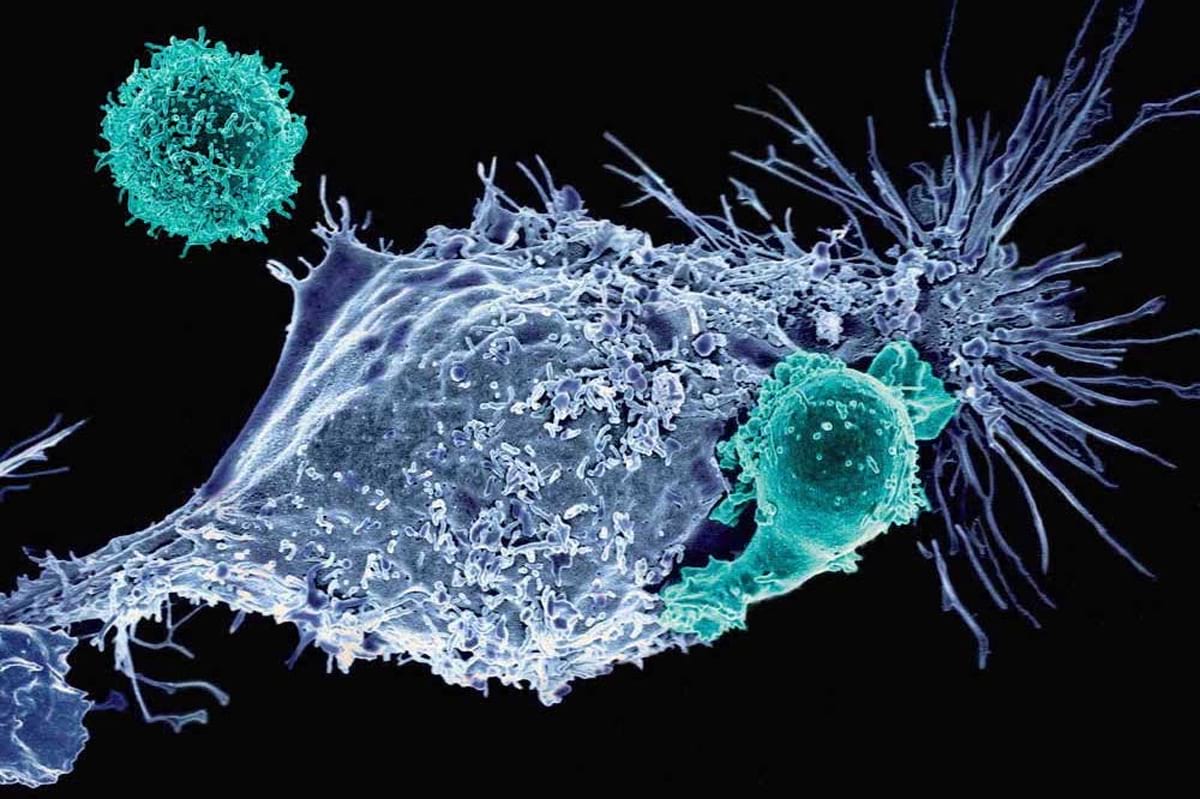
What cancers does CAR T therapy treat?
CAR T cell therapy treats several blood cancers, including:
- Relapse or refractory B-cell leukemia (B-ALL) in children and adults.
- B-cell non-Hodgkin lymphoma (B-NHL) in adults ages 18 and older.
- Multiple myeloma (MM) in adults ages 18 and older.
CAR T cells therapy is also being used in research studies, called clinical trials. We are researching the use of CAR T cell therapy for both children and adults.
Why choose MSK Kids for CAR T cell therapy?
MSK Kids is one of the world’s most experienced cancer centers in CAR T cell therapy. Our MSK Kids team uses CAR T cell therapy to treat leukemia, lymphoma, and solid tumors.
MSK scientists played a key role in developing the science and technology of CAR T cell therapy. We were involved in the research that led the U.S. Food and Drug Administration to approve CAR T cell therapy.
MSK Kids also is a leading researcher on using CAR T cell therapy to treat childhood cancers. Your child may be able to join our research studies, known as clinical trials, that use CAR T cell therapies for childhood malignancies (muh-LIG-nun-sees). A malignancy is when abnormal cells keep dividing and spread to nearby tissues. You can find information here about MSK’s clinical trials for children. Enter “CAR” or “Car T-Cell” in the search bar to find a clinical trial that may be right for your child.
At MSK Kids, our team offers the support your family needs during CAR T therapy. Families come to us from across the U.S. and around the world. MSK Kids has resources available to help you plan ahead and we can help you find local housing and plan your transportation to get to MSK. We also offer support services to take care of your child’s medical and emotional needs.
Please call 833-MSK-KIDS (833-675-5437) if you would like more information about CAR T cell therapy. Our team will answer your questions and talk with you about whether this treatment may be right for your child.
If CAR T cell therapy seems like a good option, we will ask for your child’s medical records. MSK Kids will schedule a consultation appointment with one of our doctors. We can move forward with scheduling treatment if CAR T cell therapy is right for your child.
Please read on for information about CAR T cell therapy from MSK Kids experts. We have information about how CAR T cell therapy works and what to expect during treatment.
You may have questions about how well CAR T cell therapy works. You may want information about what happens if CAR T cell therapy does not work well for your child. When you make an appointment at MSK Kids, please bring a list of questions for your doctor to answer.
Your CAR T cell therapy care team
Our CAR T care team includes experts from many MSK Kids departments. There are specialists in cellular therapy from MSK Kid’s Transplant and Cellular Therapy service. There are also experts in neurology, intensive care, pharmacy, and nursing. All team members are specially trained in caring for children and young adults during their CAR T cell therapy.
How does CAR T cell therapy work?
Video: CAR T Cell Therapy: What It Is and How It Works
This therapy makes your child’s immune system fight cancer. During CAR T cell therapy, we genetically modify (change) some of your child’s T cells to make them kill cancer cells. The treatment is like a living drug. These changed T cells can stay in your child’s body for months, even years, giving protection against cancer.
Here are the steps:
- We collect T cells (from the child/patient or healthy donor).
- These T cells are engineered in a lab to fight cancer.
- When the CAR T cells are ready, your child will have conditioning chemotherapy. We will then put the cells in your child’s blood.
- We closely monitor your child for side effects.
In the first step, we collect T cells that we will use to treat your child. We get the T cells from your child’s blood, or sometimes, from a healthy donor. This process is called leukapheresis (LOO-kuh-feh-REE-sis). For more information on leukapheresis, you can read our patient education resource CAR T Cell Therapy: A Guide for Adult Patients & Caregivers.
Most children will need placement of a special type of catheter (tube) before T cell collection. Your care team will make sure your child is relaxed and comfortable during this procedure. Collection of T cells is almost always done as an outpatient procedure. You will not have to stay overnight in the hospital.
In the second step, we send the T cells to a lab to genetically modify (change) the T cells. This is called genetic modification. Scientists put a new gene into the cell that lets them make the CAR protein. This protein makes T cells recognize and kill cancer cells. Once the T cells have this new gene, they’re called CAR T cells. It takes about 20 to 30 days for the CAR T cells to be ready.
Next, while the CAR T cells are getting ready, your child will have a cycle of conditioning chemotherapy. This chemotherapy helps the CAR T cells work better at finding and killing cancer. Once your child’s body is ready, the CAR T cells will be infused (put) into their bloodstream through an IV. This takes about 30 minutes and is similar to a blood transfusion.
CAR T cell infusions can be done safely in the hospital (inpatient) or at our clinic (outpatient). There are few side effects during or right after this procedure. For most people, this is an outpatient procedure at MSK’s clinic. Your doctor will talk with you about where your child’s conditioning chemotherapy will take place.
In the final step, your care team will watch your child closely for side effects. CAR T cell therapy is a treatment that takes a long time. You must stay near MSK after your child’s CAR T cells infusion. You must be within 1 hour of travel time to MSK for up to 30 days. Your doctor and clinical care team will tell you how often you need to come back. You will come back to either the clinic (outpatient) or stay in the hospital (inpatient).
CAR T cell therapy side effects
The CAR T cell therapy experts on the MSK Kids team are among the most experienced in the world. CAR T cell therapy has unique side effects, and they can be mild or severe. Your doctor will talk with you about which side effects are likely or rare for your child’s CAR T cell therapy.
We will do everything possible to make your child safe and comfortable during this therapy. Your care team will watch your child carefully for any side effects, and manage them. Your care team may need to admit your child to the hospital to monitor and treat side effects. MSK Kids is very experienced in managing the more severe side effects that some children may have. These side effects can include:
- A fever of 100.4 °F (38 °C) or higher.
- A faster heart rate than usual.
- A low blood pressure.
- A low level of oxygen.
- Trouble breathing.
- A change in how the kidneys work.
- A change in how the nervous system works.
Health insurance and CAR T cell therapy
CAR T cell therapy is an approved treatment for some hematologic (blood) cancers. Insurance may cover the cost of CAR T cell therapy and related care.
Your child may be having CAR T cell therapy as part of a research study, also called a clinical trial. If so, you will not have to pay for the cost of the CAR T cells. However, you or your health plan must pay for the treatment costs related to CAR T cell therapy.
Our CAR T cell program coordinators will connect you with an MSK financial adviser. They will answer your questions about paying for treatment. You can find information about insurance and financial assistance here.
MSK Care Advisors are available 24 hours a day, 7 days a week.