For every windsurfing dandelion seed that lands in a nurturing spot and sprouts a new weed, perhaps a thousand more will fall on hostile ground or be trampled under foot.
Something similar happens in cancer, which explains why one of the most famous ways of thinking about its spread is called the “seed and soil” hypothesis. First articulated by the English surgeon Stephen Paget in 1889, the idea is that the ability of a cancer to spread (metastasize) to other locations depends as much on the tissue environment the cancer lands in — the soil — as the character of the cancer cell — the seed — itself.
This could explain, for example, why breast cancers tend to metastasize to bones and the lungs; why colorectal cancer tends to spread to the liver; and why melanoma can spread pretty much anywhere.
For many years, scientists focused most of their attention on the “seed.” They looked for the source of cancer’s deadly power in the genetic changes occurring in cancer cells themselves. But that approach tended to gloss over the importance of the environment — not only local tissue conditions but also the action of the immune system.
In the past decade, research from Memorial Sloan Kettering scientists has revolutionized our understanding of both the seed and the soil — and how the two interact to promote metastasis. With this new perspective, and support from the Alan and Sandra Gerry Metastasis and Tumor Ecosystems Center (GMTEC), MSK scientists are developing innovative strategies to stop cancer’s spread.
A Long and Difficult Road
The need is painfully clear: Metastasis is responsible for 90 percent of cancer deaths. Once a cancer spreads, it’s nearly impossible to contain.
Yet this depressing observation obscures a hidden silver lining: For all its deadly power, metastasis is not something that cancer cells can do easily. Much like an individual dandelion seed, the odds of a cancer cell metastasizing are not in its favor.
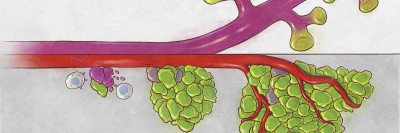
Between dislodging from a primary tumor and reaching fertile soil are many obstacles that a cancer cell must surmount. First, there is the powerful shear force that comes from hurtling through the bloodstream. Then, there’s the challenge of finding an exit from that tumultuous flume ride: a tiny gap in a blood vessel wall through which to crawl to relative safety.
And that’s just one leg of the journey. The new environment itself presents unfamiliar conditions. To survive — let alone grow and multiply — the would-be metastatic cell must be capable of adapting to its surroundings. That includes finding ways to dodge the searching sensors of the immune system.
Of course, considering the many millions of cancer cells that break off from a primary tumor every day, it’s not surprising that some do manage to clear all the hurdles. But to scientists seeking solutions, each of those hurdles represents an opportunity.
Laying Low
MSK researchers are especially keen to understand what is, perhaps, the most confounding and frightening aspect of metastasis — how lone cancer cells can sometimes survive undetected in the body for months and even years at a time without being killed, before eventually sprouting new tumors. Scientists call this period dormancy.
Dormancy is difficult to study in people, but there’s no question it occurs. The most dramatic evidence comes from people who once had cancer, were cured, and then donated an organ. The organ recipient then develops cancer — the very kind of which the donor was cured. The only possible explanation is that the donated organ contained metastatic cancer seeds that were lying dormant — perhaps kept in check by the donor’s immune system.
Several years ago, Sloan Kettering Institute Director Joan Massagué and his colleagues made a startling discovery that explains how this could happen. They found that dormant cancer cells have the ability to cycle between periods of active division and a sleep-like state called quiescence. When the cells are actively dividing, they grab the attention of immune cells called natural killer cells, which surround and kill them. But when cancer cells are sleeping, they go undetected.
The periodic cycling means that the lurking cancer cells are never killed completely, and the immune system puts pressure on the surviving cancer cells to evolve new survival traits — including, eventually, the ability to escape immune control. In alternating between periods of active division and dormancy, these rogue cancer cells resemble stem cells — a population of cells that lives in and helps repair tissues.
“Dormancy is a normal state for the stems cells in our body,” says Dr. Massagué, who holds the Marie-Josée and Henry R. Kravis Foundation Chair and is Executive Director of GMTEC. “They stay dormant until there is a need for their activity to replace lost or damaged cells.”
When cancer cells go dormant, they not only escape detection by the immune system, they also evade being killed by treatments like chemotherapy, which work only against dividing cells. It’s a surprisingly effective survival trait.
Turning Back the Clock
Just this past year, Dr. Massagué’s lab — this time in collaboration with the labs of computational biologist Dana Pe’er and physician-scientist Charles Rudin — discovered another trick that cancer cells employ to spread. By studying lung cancer in mice and in human tissue samples, they found that lung cancer cells switch on genes that are usually active only during lung development. The cells in a sense “go back in time” to an earlier stage of development as they spread from a primary location to a distant site.
Anna-Katerina “Kat” Hadjantonakis, Chair of the Developmental Biology Program in SKI and Alfred P. Sloan Chair, remembers hearing about these discoveries for the first time. “Joan called me and said, ‘We see this sequence of gene expression — does it mean anything to you?’ And I said, ‘Oh my gosh, this is the same sequence we see in an embryo as a lung forms.’”
The tumors, in effect, “reenact the developmental program for making a lung, but out of time and out of place,” Dr. Hadjantonakis says.
A key technology for making this discovery is called single-cell RNA seq (an abbreviation for sequencing pronounced “seek”). It allows researchers to measure precisely which genes are turned on or off in individual cells among thousands of cells all at once.

Dr. Pe’er, a single-cell RNA seq expert who collaborated on the study, thinks the lung cancer findings are likely to apply across different cancer types. In fact, the study, which was published in Nature Medicine in 2020, served as the basis for a large grant awarded to MSK from the Human Tumor Cancer Atlas Network, a National Cancer Institute-funded Cancer Moonshot initiative. Dr. Pe’er and physician-scientist Christine Iacobuzio-Donahue, the David M. Rubenstein Chair, are the principal investigators. They are studying the transition from primary cancer to metastasis in lung, pancreatic, and colon cancers.
These organs share something in common: They are all derived from the same layer of the embryo called the endoderm.
“Some of the nastiest cancers are those involving endoderm-derived organs,” says Dr. Pe’er, who is Chair of the Computational and Systems Biology Program in SKI, holds the Alan and Sandra Gerry Endowed Chair, and is Scientific Director of GMTEC. “For combating tumors derived from these organs, understanding normal development is going to be critical.”
That’s why she and Dr. Hadjantonakis are collaborating on a project to understand the development of endoderm-derived tissues.
Dr. Pe’er and her colleagues are also looking at metastasis to the brain, which is dramatically different than other organs. “The brain is as foreign an environment as a pancreas or lung cancer cell could possibly find itself in,” Dr. Pe’er says. “The degree of adaptation that these cells must undergo is extreme.”
With physician-scientist Adrienne Boire, Dr. Pe’er is using single-cell analysis to explore these adaptations. They’ve already identified several that could be potential therapeutic targets.
Switching Tracks
The shape-shifting quality that metastatic cancer cells exhibit may also explain some cases of drug resistance. Several years ago, lung cancer specialists Helena Yu and Dr. Rudin began to notice that some of their patients, when treated with drugs called epidermal growth factor receptor (EGFR) inhibitors, eventually developed resistance to these targeted drugs. But the way the tumors became resistant was unlike anything they had ever seen. The cells transformed into an entirely different type of lung cell that is not dependent on the growth receptor at all.
“It’s as if the cell gets off one set of train tracks and jumps to a completely different set,” says Dr. Rudin, the Sylvia Hassenfeld Chair in Lung Cancer Research at MSK. “In order to make that switch, the cells have to first go backward toward a less differentiated state. And to do that, they resort to some of the same developmental genes that we also see being turned on in the context of metastasis.”
As Dr. Rudin and other MSK scientists are coming to learn, this identity switching is not restricted to lung cancer. Almost exactly the same switching is seen in prostate cancer that is being treated with antiandrogen therapy and in breast cancer that is treated with antiestrogen therapy.
The findings have already changed clinical practice. Now, when someone relapses, doctors routinely re-biopsy the tumor to find out if it has changed. If it has, then they know to switch the treatment to match the new type of cancer. Clinical trials are underway to test drug combinations to prevent drug resistance.
The goal of MSK researchers is to turn metastatic cancer into a manageable chronic condition and, perhaps one day, uproot cancer for good.
Outbreak
After dislodging from a tumor, after going dormant, after trying on different identities, cancer cells must surmount one final hurdle to spread: They must acquire properties that allow them to grow in a new territory.
Tracking this last step has been the focus of physician-scientist Karuna Ganesh, a Josie Robertson Investigator. In the past year, her lab found that a particular molecule, called L1CAM, appears to be required for metastatic cells to grow in a new location. L1CAM was originally identified in the context of brain development. But Dr. Ganesh’s research has shown that it also plays a crucial role in both colon repair and in colon cancer.
In a normal colon, activating L1CAM in tissue stem cells is part of the way that the colon repairs itself after an injury such as colitis. Stem cells making L1CAM can separate from their neighbors, migrate into the gap of a wound, and then start to fill it in with healthy new cells.
But L1CAM also permits metastatic cancer cells to repair the tumor — and regenerate it in a new location. In fact, cancer cells can’t grow without it. The researchers can see this when they knock out L1CAM in tumor cells growing in mice. The cells are able to seed new areas, but they do not germinate. With L1CAM, however, the cells begin to crawl along blood vessels and make themselves at home in their new location, eventually forming a new tumor. Based on this insight, Dr. Ganesh’s lab is actively looking for drugs that could block L1CAM.

The fact that metastatic cancer cells co-opt the body’s wound-healing system is devilish but also makes a kind of twisted sense. A wound is essentially a hole in a tissue, which is very dangerous for an organism. To repair this hole, tissues rely on stem cells that turn on a wound-healing program. Because cancer creates a wound where it grows, it causes malignant cells to adopt this same wound-healing behavior. It is these cells that will regenerate the tumor.
“Metastasis is wound healing gone wrong,” Dr. Ganesh says.
By learning more about the ways that cancer cells repurpose these and other properties of stem cells, researchers hope to throw up barriers to metastasis.
“My dream is that the research being done now on dormancy and outbreak will lead to therapeutic breakthroughs,” Dr. Massagué says. “One possible approach might be to trick the dormant cells into expressing those genes that alert the immune system so they are eliminated the same way that 99 percent of their comrades were eliminated.”
He hopes that learning more about the steps that lead to metastasis in the first place will also help physicians understand what happens when cancers relapse.
Over the next decade, with the help of sophisticated new technologies, MSK researchers plan to mount a concerted attack on the problem from all sides. Their goal is to turn metastatic cancer into a manageable chronic condition and, perhaps one day, uproot cancer for good.