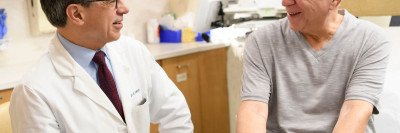
Genetic counseling and cancer risk assessment is an important part of cancer care at Memorial Sloan Kettering. Through this process, our genetic counselors and doctors will guide you through the process of learning about your risk for inherited cancer. Genetic counselors are experts in collecting and assessing information about family history.
Based on genetic test results and family cancer history, the Clinical Genetics Service (CGS) provides patients and family members with recommendations for surveillance and risk reduction. These recommendations are provided in a compassionate and easy-to-understand manner.
CGS’s aim is to provide people who have cancer and their family members with compassionate care. We help people understand how a diagnosis of cancer in one family member may impact the health of others. Knowing that a genetic predisposition to cancer exists in a family can help identify other relatives who are at risk for the disease. This knowledge can lead to cancer prevention and early intervention for entire families.
If you have been diagnosed with cancer or have a family history of cancer, you may be interested in learning more about genetic counseling and the options for genetic testing. Genetic counseling is often recommended for people who develop cancer at a young age (for example, breast cancer before age 45). It may also be recommended for people with multiple close family members who have had the same type of cancer. We take many risk factors into consideration when evaluating someone for a potential hereditary risk of cancer.
If you are interested in learning about a potential hereditary risk of cancer, we encourage you to read through this guide and contact our Clinical Genetics Service.